DMEM salt/buffer concentrations
I talked about this yesterday in lab meeting, but here is the data. Since the components of HBSS by themselves did not cause discoloration (HBSS components), I adjusted the concentrations of NaCl, KCl, glucose, Na2HPO4 and NaHCO3 to match that of DMEM. The control was a wafer in DMEM/F12 with 10% FBS. None of these samples were RTP’d.
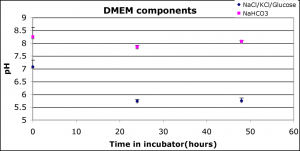
The KCl concentration is the same in DMEM and HBSS, the NaCl is lower in DMEM than HBSS and the glucose and NaHCO3 are both higher in DMEM than HBSS. NaHCO3 is about 10 times greater in DMEM than in HBSS. Only NaHCO3 caused discoloration in this experiment. Here’s how the pH changed over time:
The pH of the NaCl/KCl/Glucose solution closely matched the NaCl/KCl and Glucose pH’s from my previous post. However, the pH of NaHCO3 did not decrease as much as in previous experiments, so it’s hard to conclude if discoloration is pH- or solution- mediated.

The patterns of discoloration are strange. Are the solutions stirred in the incubator? It seems unlikely that the material would be different in the center than the outside, so there is some environmental effect going on. The right column is particularly concerning since you have areas that are completely discolored, while most of the area is untouched. Is it possible that local variations in pH or concentration are developing? Are there bubbles or any other observable chemical/geometrical effects?
Yeah, I was surprised by that, too. It seemed to stop discoloring. I thought that maybe something adsorbed around the edges and prevented discoloration. I suppose there could be local effects – I don’t mix the solutions in the incubator. I’ve never noticed any bubbles, but every once in a while we see strange discoloration patterns.