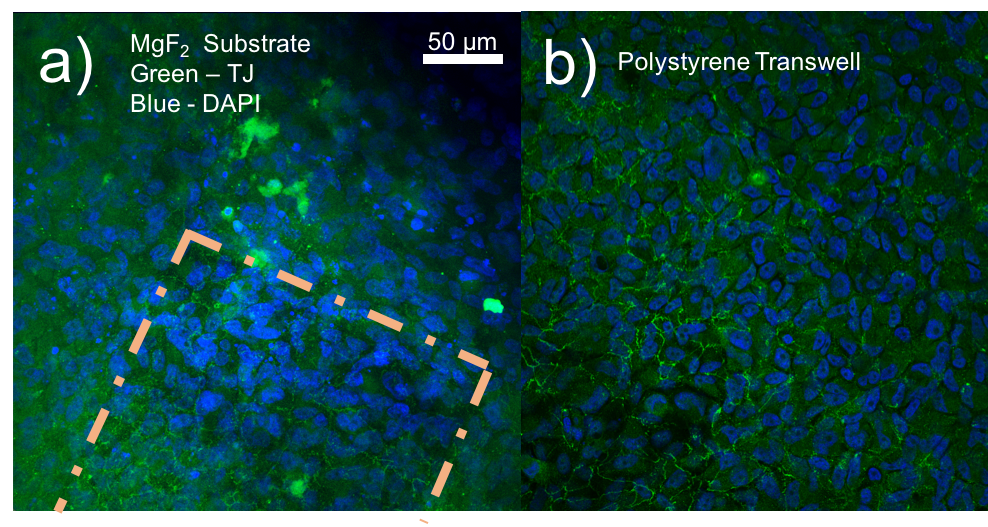
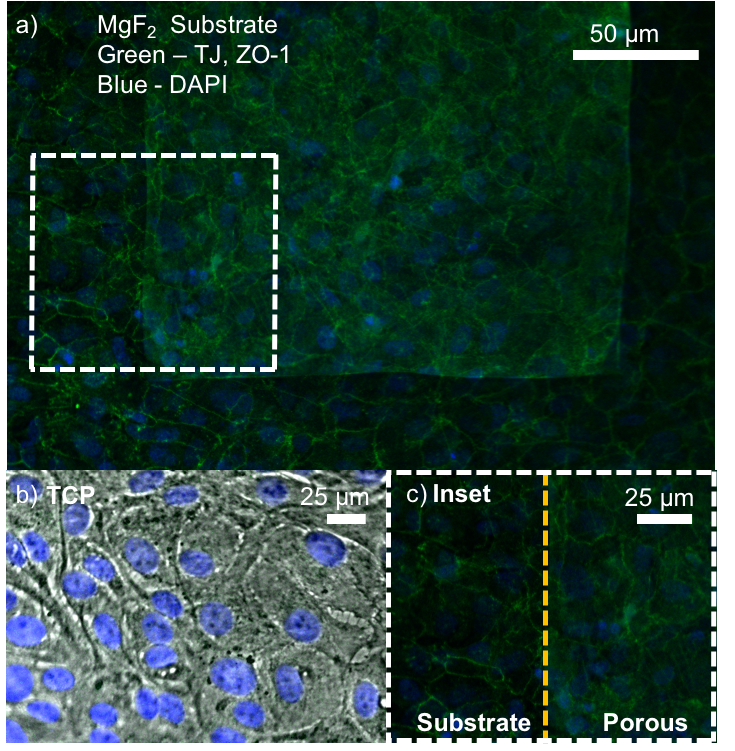
ZO-1 staining of confluent RPE cells on various MgF2 substrates
I’ve been culturing RPE cells over here in Rochester for my MgF2 fabrication paper, in which the goal is to make a permeable Raman compatible cell substrate. My previous work had shown polarization of RPE cells (via-ZO-1 staining) on nanoporous MgF2 nanomembranes, but in the meantime, I have reworked my MgF2 substrates to be thicker and stronger, with larger 400 nm pores. I cultured P6-P8 cells on a variety of substrates (high porosity chips (1:1 spacing), glass coverslips coated with MgF2, silicone, TCP) for 22 days. These were then fixed and stained for ZO-1. None of these membranes broke during culture.
Previous Results
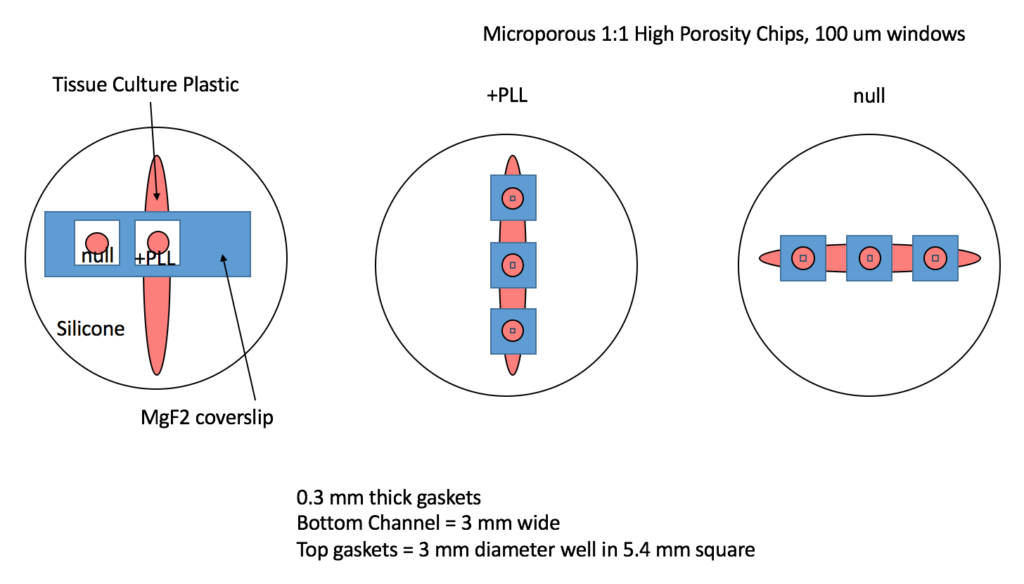
Treatments
- TCP
- null
- Microporous MgF2 (2o0 nm thick, High Porosity, 1:1 400 nm pores, 100 micron square windows)
- + .01% PLL
- null
- Coverglass coated with MgF2 (200 nm thick, not permeable)
- + .01% PLL
- null
- Silicone (cells that spill over onto the silicone gaskets)
- null
Devices
Procedures
- 1/9/17
- Devices were autoclaved
- Seeded P6 RPE cells at 5000 cells/mm^2 in flat devices
- wanted about 50 cells in 100 um square window, resulted in 35,325 cells/well,
- Observed about 30-40 cells in a window immediately after seeding
- Cells were fed 3x a week
- RPE Complete Media = DMEM + sodium pyruvate + L-glutamine + 10% FBS
- Imaged 1x a week (phase)
- 1/18/17
- Cells on the MgF2 coverslip all died. Reseeded P8 RPE cells on the coverslip after washing with 1x PBS.
- 2/1/17 – Fixation and staining
- Aliquot Antibodies
- F0382 SIGMA Secondary antibody: Anti-Rabbit IgG (whole molecule)–FITC antibody produced in goat affinity isolated antibody, buffered aqueous solution
- use at 1:80-1:50 dilution
- Aliquot 2ML into 50x 40UL portions
- to Make 2000uL of stain (1:50), add aliquot to 1960ul of solution (1%BSA in PBS)
- HPA001636 SIGMA Primary Antibody: Anti-TJP1 antibody produced in rabbit Ab1, Prestige Antibodies®Powered by Atlas Antibodies, affinity isolated antibody, buffered aqueous glycerol solution
- Use in 1:200 dilution for immunohistochemistry
- Aliquot 100uL into 10x 10uL portions
- To make 2000uL of stain, add aliquot to 1990 uL of solution (1%BSA in PBS)
- This is enough stain for 2000uL/150uL = 13 devices
- F0382 SIGMA Secondary antibody: Anti-Rabbit IgG (whole molecule)–FITC antibody produced in goat affinity isolated antibody, buffered aqueous solution
- Prepare Solutions
- 1% Triton X-100 in PBS
- 4% PFA in RPE Media
- 2mL of Primary antibody
- 2mL of secondary antibody (following day)
- Fix Cells (22 days)
- Wash devices with 1x PBS
- Fix with 4% PFA in warmed Cell media (37C) for 30 minutes in fume hood
- Wash Devices with 1x PBS 3 times
- Permeabilize + Stain Cells
- Permeabilize with Triton X-100 (0.1% in PBS) for 15 minutes at room temperature
- Wash with 1x PBS
- 1% BSA in PBS for 1 hr to block aspecific binding
- Wash with 1x PBS 2 times
- Add Rabbit-Antihuman ZO-1 antibody (150uL of 1:200 diluted antibody, 10 ug/uL) to device, and incubate overnight in the fridge
- The following day, wash with 1x PBS 3 times
- Add secondary antibody FITC-antigoat (150 uL of 1:50 diluted antibody) for 1.5hrs
- Wash with 1xPBS 3 times and submerge devices in PBS
- Store in Fridge
- Aliquot Antibodies
Mounting and Imaging
I removed the silicone gaskets surrounding each of wells and flipped over the devices to image in a small dish (immersed in 1x PBS).
Results



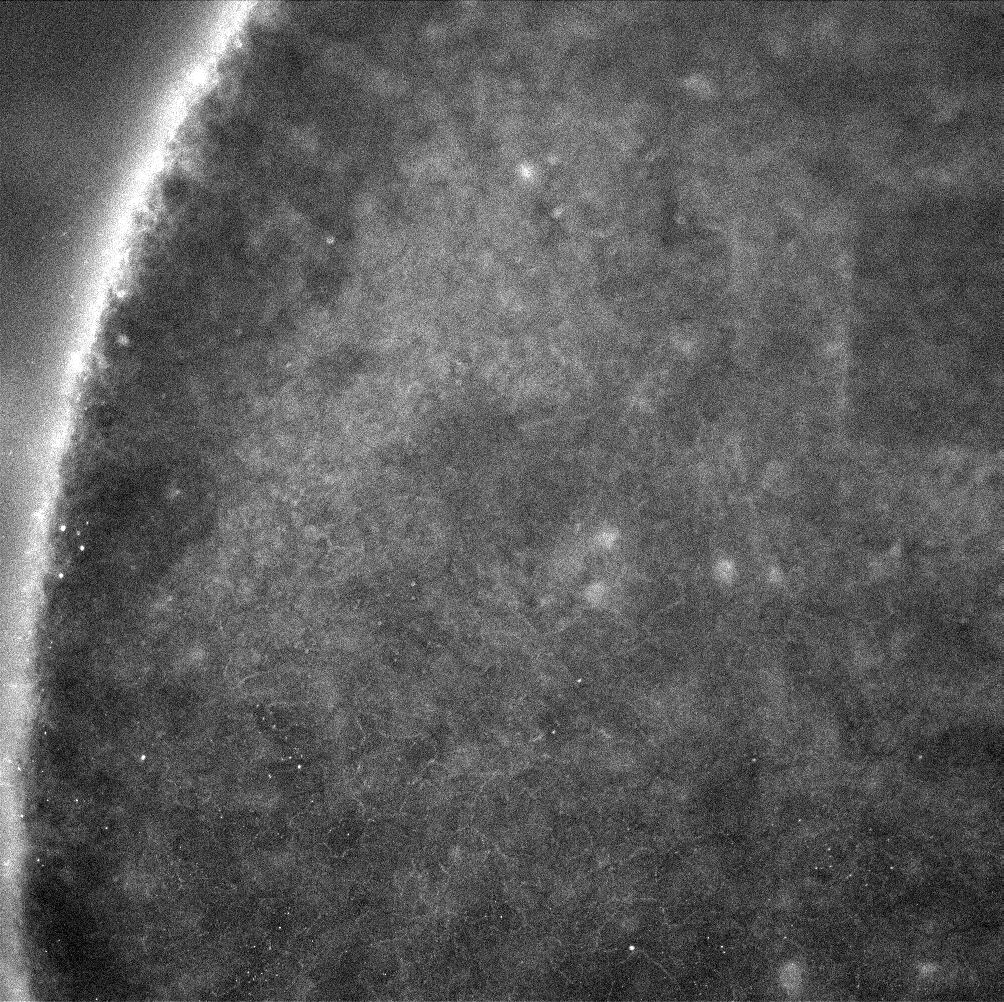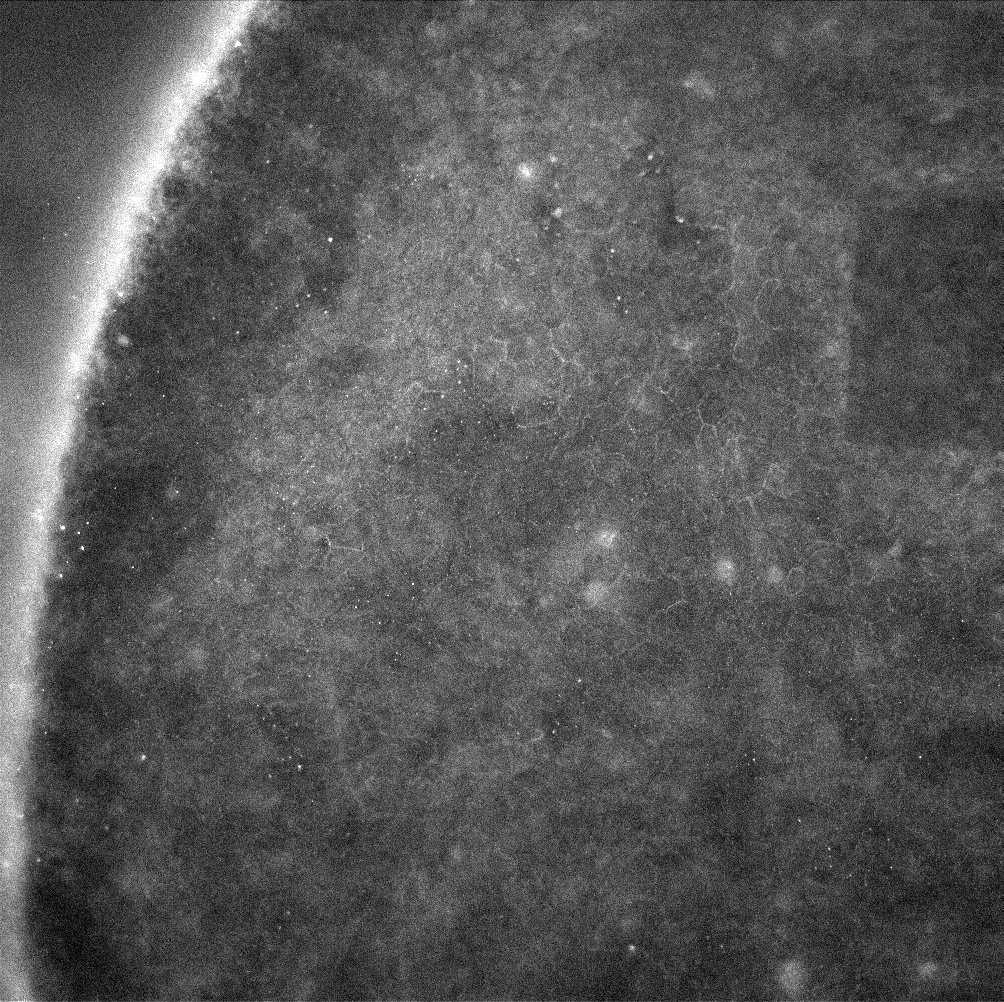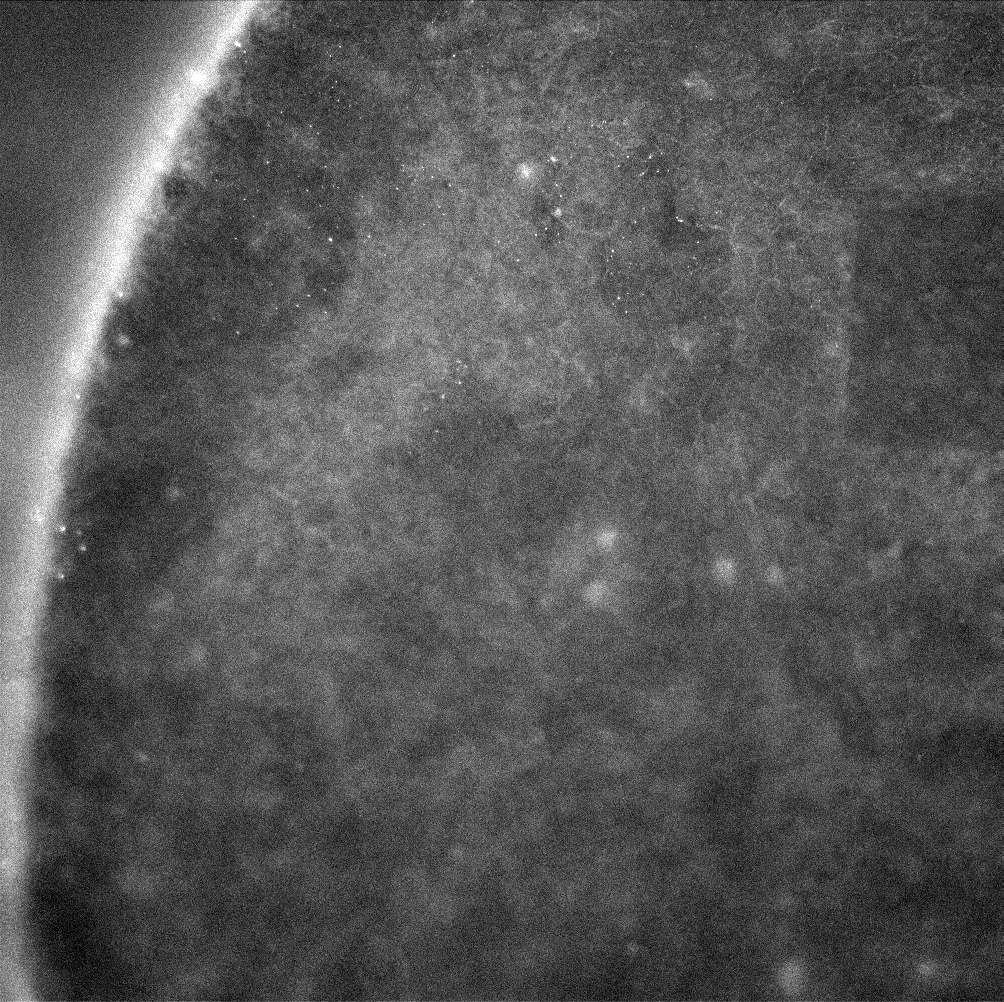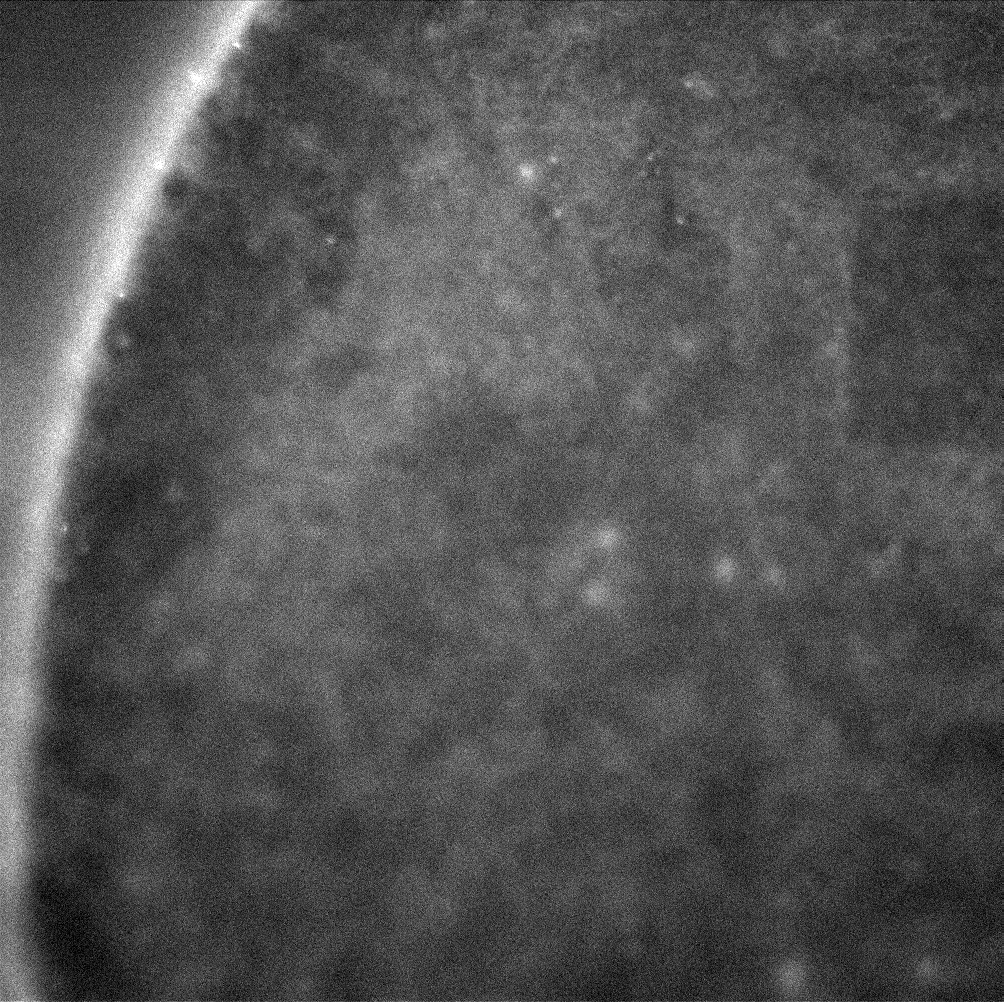

Future
I believe my washing steps could be better. The amount of fluorescence is pretty low in all of these images. I am also going to use larger gaskets that can access more media at once.
