Maintaining Polarized Inflammation and ICAM-1 mAb Blocking – FINAL Polarized TNF-alpha Revision Experiments
Introduction
Read the manuscript here: Integrative Biology Publication
Please refer to the linked manuscript for a detailed introduction. In short, we have been interested in characterizing the role of endothelial cell (EC) apicobasal polarity in inflammation. While usually reserved for the epithelium, ‘apicobasal polarity’ is a term describing a monolayer or cell with defined apical and basal interfaces. Recent evidence suggests the ultrathin endothelium may have inherent apicobasal polarity as well. We begin this investigation by understanding how EC apicobasal polarity may direct immune responses in inflammation originating in the tissue versus the blood. This work, now published in Integrative Biology, was updated in a recent set of revision experiments. The reviewers wished to better understand two main findings/methods in the paper: 1) How is the polarized inflammatory stimulation maintained over the 24h time point? and 2) Can the involvement of ICAM-1 in the model of local inflammation be further characterized? The experiments below constitute our response to these major comments.
Relevant Blog Posts
Introduction Blog Post w/ Majority of Figures
Initial Revisions – 10&70 kDa Dextran Pe Blog Post
Methods
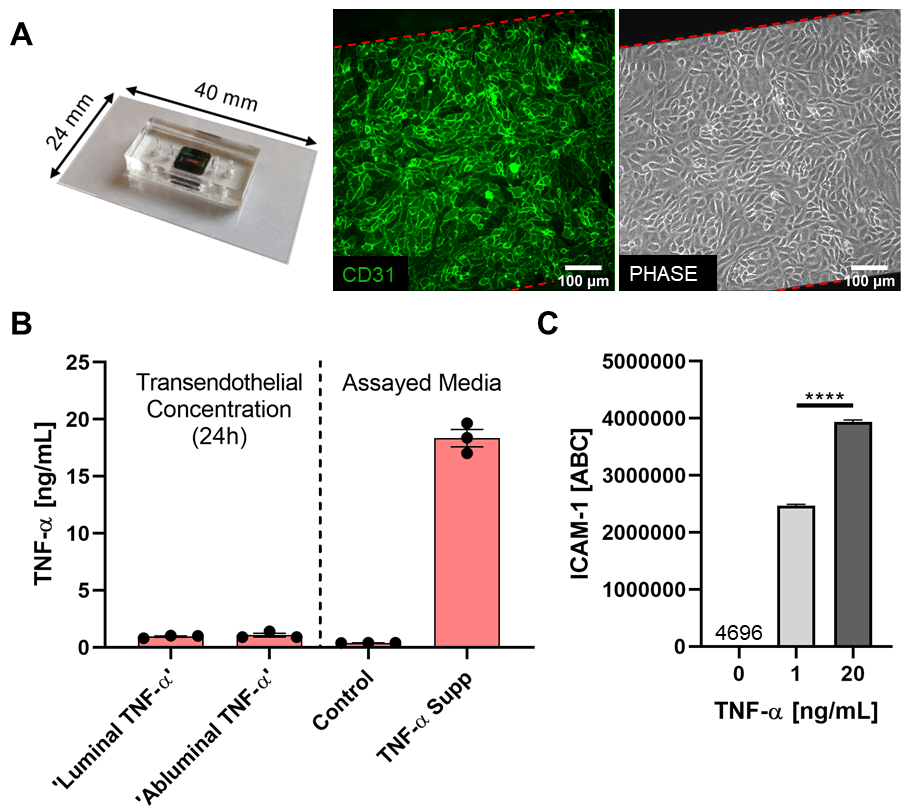
TNF-α Transendothelial Flux
The μSiM-MVMs were perfused luminally or abluminally with TNF-α supplemented media [20 ng/mL] and incubated for 24h. Media was gently collected from the trans-channel following the 24h incubation (i.e. opposite channel to which TNF-α was added at hour 0) and TNF-α content was quantified by ELISA (Human TNF-alpha Quantikine ELISA; R&D Systems, Minneapolis, MN). Media (both control and TNF-α supplemented) added at hour 0 was assayed in parallel.
ICAM-1 Flow Cytometry
HUVECs were seeded and grown to confluency in tissue culture-treated 6-well plates. Confluent HUVEC monolayers were incubated with 0, 1 or 20 ng/mL TNF-α for 24h. Stimulated HUVECs were trypsinized and incubated on ice with anti-human ICAM-1 monoclonal mouse IgG1 conjugated to Alexa Fluor 488 (R&D Systems, Minneapolis, MN). In parallel, Quantum Simply Cellular anti-Mouse IgG beads (Bang Laboratories Inc., Fishers, IN) were incubated with saturating concentrations of the same conjugated antibody. Geometric mean fluorescence intensity was determined by flow cytometry (Guava Easycyte Mini; Luminex, Austin, TX). Geometric mean intensity was converted to antibody binding capacity (ABC) by establishing a standard curve using the Simply Cellular beads. This conversion allowed for direct quantification of ICAM-1 expression on the HUVEC surface.
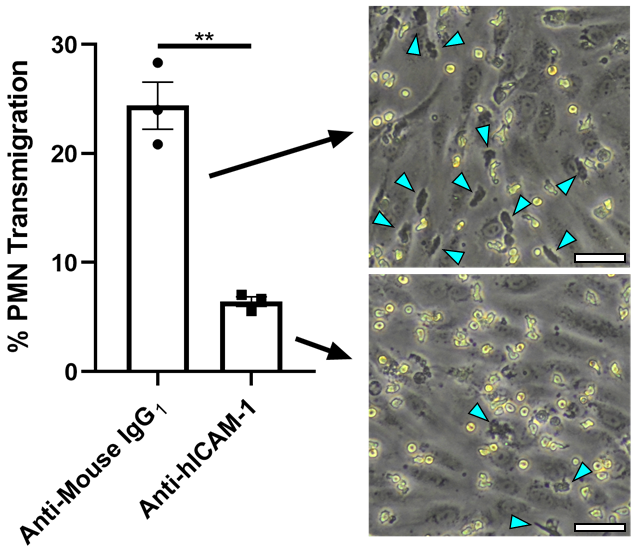
ICAM-1 Blocking & PMN Transmigration
In ICAM-1 blocking experiments, function blocking anti-hICAM-1 or isotype control antibodies (R&D Systems, Minneapolis, MN) were added to the luminal channel of abluminally stimulated μSiM-MVMs (TNF-α [20 ng/mL] added 24 h prior) and incubated at 37 °C, 5% CO2 for 30 mins. PMNs were added to the luminal channel in antibody-containing media and images were recorded over 30 min. % Transmigration was quantified by dividing the total PMNs in the first image to the total number of observed transmigration events over 30 min.
Membranes:
- 100 nm NPN, Wafer #1349(?)
Results
In order to assess the HUVEC barrier to TNF-a passage, experiments were performed as usual, and media was collect from the trans-compartment (i.e. if media was added luminally, abluminal media was collected for ELISA). HUVECs existed as a tight barrier to TNF-a passage, with < 1 ng/mL TNF-a detected in the trans-compartment, regardless of side first receiving the stimulus (Figure 1B). To confirm that 1 ng/mL and 20 ng/mL were not both saturating concentrations of TNF-a, ICAM-1 surface expression was assessed following 24h incubation with the respective concentrations. While both concentrations induced a substantial inflammatory response, HUVEC ICAM-1 surface expression was higher when stimulated with 20 ng/mL versus 1 ng/mL, thus HUVECs can differentiate the two concentrations (Figure 1B). These results ultimately ensure we are maintaining a polarized inflammatory environment over the course of our experiments.
Further characterization of ICAM-1 involvement of PMN transmigration in our model of local inflammation (abluminal TNF-a) was needed to support our conclusions. Thus, we opted to blcok ICAM-1 on the surface of abluminally stimulated HUVECs and repeat transmigration experiments. When compared to an isotype control, we observed a statistically significant decrease in PMN transmigration across blocked HUVEC monolayers. We conclude that, in combination with our confocal data, ICAM-1 is an important player in transmigration, warranting further experimentation.
Conclusions
TNF-a passage is limited across confluent TNF-a monolayers within our system. Additionally, ICAM-1, potentially through surface reorganization, may play a more prominent role in the immune response to local infection and requires more study.