Monolayer Culture of mouse CMECs on μSIM A-Line Devices
Introduction
In the Glading Lab we study a rare disease called Cerebral Cavernous Malformations (CCMs). In this disease, brain capillaries develop greatly enlarged lumens and a disrupted blood-brain barrier. A genetic cause of the disease is the presence of a mutation in KRIT1 that causes loss of KRIT1 protein function in the cells. KRIT1 binds to Rap1, which plays a role in the integrity of endothelial junctions and will lead to a leaky endothelial barrier if disrupted. Our interest is to use the μSiM devices to study mouse cerebral microvascular endothelial cells (mouse CMECs) from KRIT1 knock-out mice in vitro to investigate the formation of leaky endothelial barriers. These animals develop CCM-like cavernous lesions and we then isolate the CMECs from these animals.
The average number of cells that are isolated and pooled from 3 adult animals is around 1×10^6 cells, which can be a limiting factor in standard 2-D assays. The μSiM allows for a larger number of assays to be run from one isolation.
Methods and Results
Attempt 1A: Determine cell density for culture on devices
I needed to establish culture conditions for a monolayer of the mouse CMECs on the NPN membrane.
The coating solution used for the mouse CMECs is a 1:1 ratio of collagen IV (COL IV) to fibronectin (FN), in grams, collagen IV is the major component of the basement membrane.
I seeded the cells at varying cell densities/device (5k, 10k, 15k, and 20k cells) to try to find the number of cells that would become confluent in 24 hours.
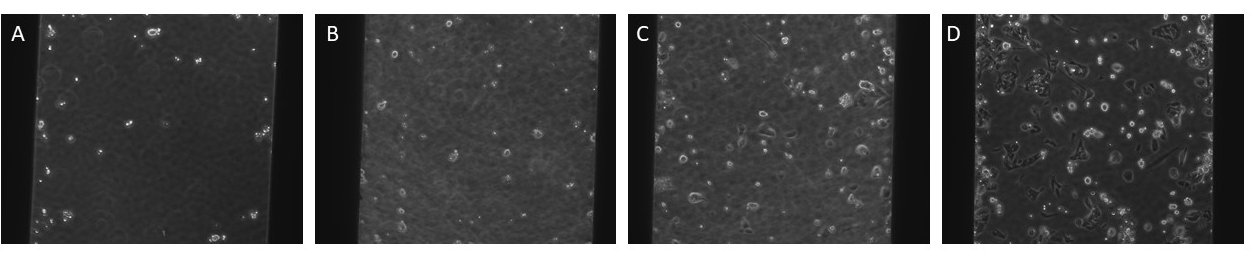
Figure 1: Representative images of the first experiment with varying cell densities/device with the membrane in the trench-up orientation 24 hrs after seeding.
A: 5k cells, B: 10k cells, C:15k cells, D: 20k cells
The cells were not confluent and they did not attach to the membrane well at these cell densities.
Attempt 1B: Determine cell density for culture on devices
I continued to increase the cell densities until I found densities that were confluent 24 hrs after seeding.
Because the permeability assay will be run ~24 hours after seeding, the monolayer needs to be confluent at this time point.
Cell densities were increased to 27.5k, 30k, 32.5k, and 35k cells.
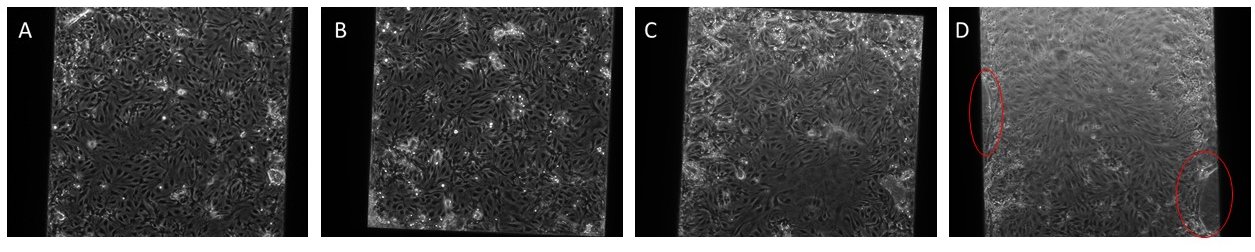
Figure 2: Representative images of the varying cell densities/device with the membrane in the trench-up orientation.
A: 27.5k cells, B: 30k cells, C:32.5k cells, D: 35k cells; Red circles in panel D show areas where the matrix/cells seem to be lifting up from the edge of the trench.
There didn’t seem to be much of a difference between these different cell densities, so I chose 32.5k cells for the next experiments.
Due to the areas in red in the images above, where the cells and matrix seemed to be “peeling” away from the sides of the trench, it was recommended that I try to seed the cells on the devices in the trench-down orientation for a flat monolayer; the McGrath lab is currently working on the development of a permeability assay for the trench down orientation.
Attempt 2: Determine culture conditions in trench-down orientation
Coat devices with FN/Col IV solution
Seed cells at varying densities/device: 25k, 27.5k, 30k, 32.5k cells

Figure 3: Representative images of varying cell densities/device with the membrane in the trench-down orientation with a new lot of NPN membrane (1380)
A: 25k cells, B: 27.5k cells, C:30k cells, D: 32.5k cells
It was interesting that the cells were not attached or as dense as I had previously seen in the trench up orientation with similar densities. My conjecture was that there was something different about the new lot of membranes or between the surface interacting with the cells on each side of the membrane. However, a member of the McGrath lab had been successful in culturing HUVECs with the same lot of membranes in the same orientation. Another difference in my protocol was that I used a different coating solution, collagen IV and fibronectin instead of collagen I and fibronectin; I had also been using a lower amount of fibronectin in the coating solution than recommended, the recommended amount is 5 µg/cm^2 or 37.3 µg/mL and I was using 1.34 µg/cm^2 or 10µg/mL.
Therefore, I increased the amount of fibronectin in my coating solution to the amount recommended and adjusted my ratio of fibronectin to collagen IV.
Attempt 3: Increase the total amount of fibronectin/collagen IV used to coat the device

Figure 4: Representative images of varying cell densities/device with the membrane in the trench-down orientation with the same lot of NPN membrane (1380).
The amount of fibronectin and collagen IV used to coat the devices was increased to match the recommended amount of fibronectin/surface area.
The top images (1) are the cells in the μSiM device and the bottom images are the corresponding coating with cells in a well in a 96-well plate, which has a similar surface area as the A-Line devices.
A: 27.5k cells, B: 30k cells, C:32.5k cells, D: 35k cells
In this experiment, compared to the experiment in Figure 3, the cells are more confluent and better attached to the membranes.
I chose to include 35k cells again in case the cell count was a part of the anomaly in the previous experiment.
However, in each of the top and bottom matched images, the density does not match the density seen in the cells cultured with the same coating solution in a well of a 96-well plate that has a similar surface area to the A-Line μSiM device.
Attempt 4: Test ECM coating solutions
I decided to test different compositions (fibronectin, collagen I, collagen IV) of the coating solutions to see if the cells had better attachment/confluency.
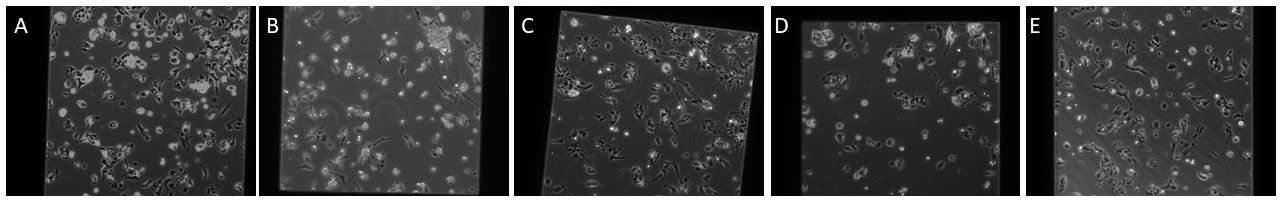
Figure 5: 35k cells seeded onto NPN membrane with varying coating solutions
A: FN, B: COL I, C: COL IV D: FN/COL I, E: FN/COL IV
It appears that the best substrate is either fibronectin or fibronectin + collagen IV
Attempt 5: Adjust the pH for fibronectin/collagen IV coating solution
By checking the pH of the coating solutions, I realized that even though the Collagen IV is diluted in PBS, the pH of the coating solutions containing Collagen IV were acidic (below 5); while the other coating solutions were at or above 7.
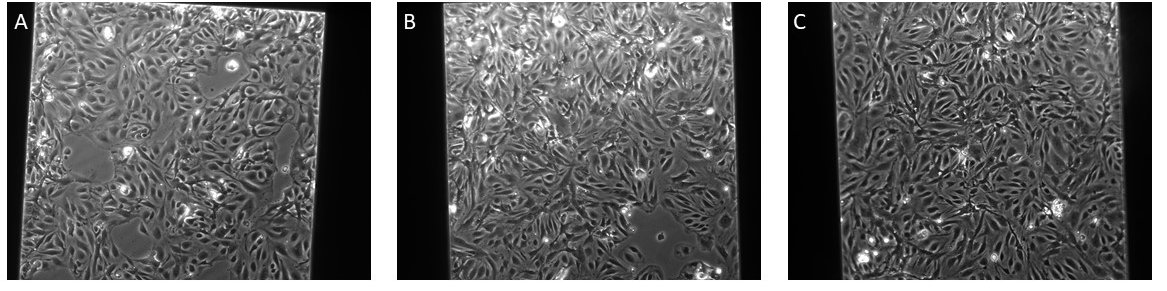
Figure 6: 35k cells seeded in the μSiM devices on NPN with different matrix coating solutions all adjusted to pH 7
A: FN, B: FN/COL I, C: FN/COL IV
By adjusting the pH of the Collagen IV and fibronectin solution to 7, I achieved a confluent monolayer of CMECs.
Attempt 6: Test culture conditions for CMECs on dual-scale membranes
CMECs were seeded at 35k cells on the dual-scale membranes with multiple coating solutions previously mentioned, pH adjusted to 7, if needed.
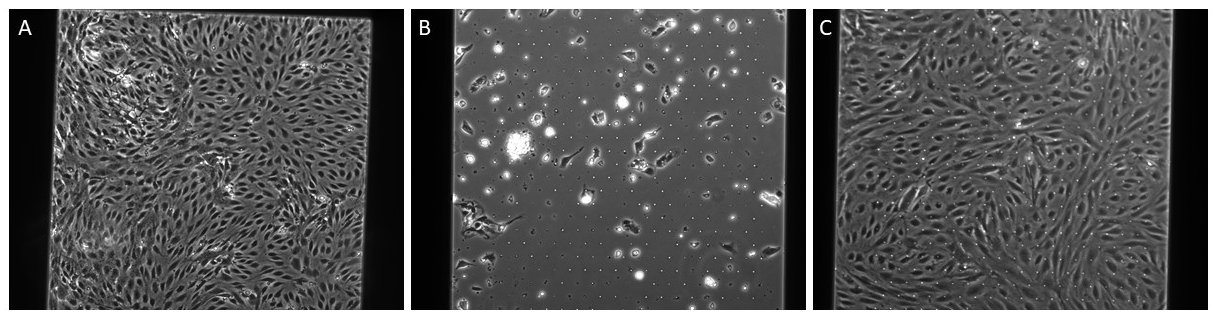
Figure 7: A: 35k cells seeded on NPN and FN/ColIV coating, B: 35k cells seeded on dual-scale membrane at 24 hours, C:35k cells on dual scale membrane at 7 days, both B and C are coated with FN only.
I was able to successfully seed CMECs on the dual-scale membranes.
Conclusions
The best culture conditions for mouse CMECs on the devices are: FN/Col IV coating solution at a concentration of 37.3 µg/mL, 35k cells per device, trench down with a pH at or above 7. I’m interested in exploring the use of the dual-scale membranes for future studies related to my project in developing a 3-D in-vitro model of CCM.