Modification of Ultrathin Microporous Silicon Nitride Membrane Stiffness for Tissue Barrier Modeling
Introduction:
Developing innovative therapeutics and treatment approaches with improved effectiveness and safety necessitates the use of cell co-culture models and barrier environments that mimic in vivo conditions. These models involve culturing two or more cell types together in vitro, allowing researchers to study cellular interactions, signaling pathways, and responses that closely resemble those seen in living organisms. In order to recreate the structural and functional properties of the extracellular matrix (ECM) found in vivo, the basement membrane (BM) plays a crucial role in these models. The BM, a specialized ECM structure underlying epithelial and endothelial tissues, provides support, regulates cell adhesion, and influences cellular behavior [1]. Notably, BM stiffness is vital for modulating gene expression in response to mechanical cues, mediated by connections between the cytoskeleton and ECM components via adhesion receptors [2]. However, achieving a substrate that closely resembles the basement membrane (BM) poses a significant challenge. The BM, which typically ranges from approximately 20 nm to 3 μm in thickness [3], is primarily composed of ECM proteins like type IV collagen, fibronectin, and laminin, with a Young’s modulus typically falling within the range of kPa (~500 kPa) as reported in numerous studies [4]. Additionally, it must facilitate physical and biochemical communication between cells, a crucial aspect for accurately replicating physiological functions. To address the latest issue, in vitro models such as barrier and co-culture setups require a porous membrane or scaffold that replicates the intricate microenvironment found in vivo [5].
Polydimethylsiloxane (PDMS), polyethylene terephthalate (PET), polytetrafluoroethylene (PTFE), or polycarbonate (PC) are widely used as synthetic basement membrane (BM) materials in constructing tissue barrier interfaces for organ-on-a-chip systems [5], owing to their advantageous mechanical properties such as flexibility, elasticity, softness, biocompatibility, and optical transparency [6]. Pore fabrication can also be achieved through techniques like soft lithography or track etching. However, while there have been advancements in developing synthetic membranes with sub-micron thicknesses that closely resemble BM, typical synthetic membranes used in organ-on-a-chip systems are often much thicker (∼10 μm) than native BMs [7]. Biocompatible hydrogels enriched with natural BM protein have also been developed to resemble native BMs. Components such as collagen I, laminin, fibrin, alginate, and hyaluronic acid are incorporated into these hydrogels to create matrices that closely mimic the BM environment [3]. Despite the versatility of synthetic hydrogels in tuning material properties such as stiffness, porosity, shape, and the controlled incorporation of adhesive ligands and degradable sequences, hydrogel BM mimics still lack certain essential characteristics of native BMs such as the minimum achievable free-standing hydrogel thickness currently ranging around 10 μm, which is at least 10 times thicker than native BMs [8].
In this project, our objective is to soften the microporous membranes to create a cell-relevant environment. To achieve this goal, Polyethylene Glycol (PEG) chains were chosen as a pivotal component of our coating structure due to their versatile surface configurations (Figure 1). When tethered to surfaces, PEG forms a distinguished ‘polymer brush’ configuration, offering a range of physical and mechanical properties that can be finely tuned to meet specific application needs by manipulating polymer chain length and density. Furthermore, deliberate bio-conjugation of the PEG polymer with selected bioactive materials results in a configuration with targeted biological properties, thereby expanding its applicability and functional capability.

Method:
Surface Characterization:
First, a microporous silicon nitride membrane with a 0.5 μm pore size underwent oxygen plasma treatment to enable the electrostatic interaction of PLL-g-PEG-Biotin on the surface. To optimize the attachment process, the membrane was subjected to oxygen plasma treatment for varying periods of time. Immediately afterward, 20 μL of 0.5 mg/mL PEG solution was introduced dropwise onto the surface and incubated at room temperature for 75 minutes. The PLL-g-PEG-Biotin concentration and the incubation time had been previously investigated [9]. The presence of the coating on the surface was quantified labeled using Alexa Fluor™ 488 streptavidin, a high biotin-binding affinity agent. A solution containing 5 μg/mL of the dye was introduced to the biotinylated surface, while a non-labeled coated surface served as the negative control. The optimum concentration of the green fluorophore label had been investigated prior to the experiment. Additionally, the surface roughness was assessed using Atomic Force Microscopy (AFM) measurements, and the contact angle was measured to evaluate surface hydrophobicity. AFM imaging was performed in tapping mode with a scanning area of 20 μm x 20 μm, and the root mean square (RMS) roughness was calculated from the acquired height profiles. Contact angle measurements were conducted using a goniometer with deionized water droplets, and the advancing contact angle was recorded as an indicator of surface wettability. All measurements were performed under the same experimental conditions to ensure comparability. A quantitative analysis was conducted using scanning electron microscopy (SEM) to examine the porosity condition of the coated membrane, and a non-coated membrane was employed as a negative control. The pores diameters were measured using ImageJ [10].
Thickness Measurement:
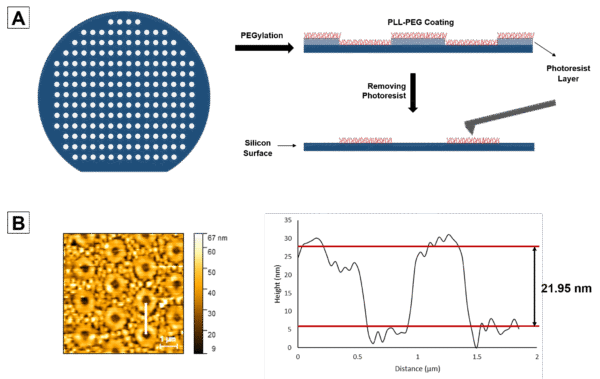
Measuring the thickness of a thin layer presents challenges. The purpose of this experiment is to measure the thickness of the PLL-PEG-Biotin coating using AFM. Initially, the silicon wafer is patterned using positive photoresist, followed by coating with PLL-PEG-Biotin, and subsequent removal of the photoresist using acetone. Atomic Force Microscopy (AFM) imaging is then conducted on the PLL-PEG-Biotin-coated silicon wafer surface in a phosphate-buffered saline (PBS) environment. A 2D height image of the surface is acquired to visualize the topography and features of the coated pattern. A height profile is generated along a line drawn across the patterned region to measure the height difference between the PEGylated spots and non-PEGylated parts of the pattern.
Cell Adhesion:
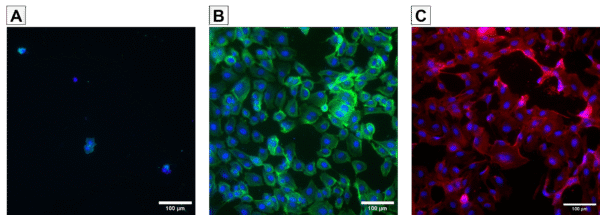
To create a cell-adhesive surface, streptavidin conjugated with fibronectin was utilized to modify polyethylene glycol (PEG), a non-fouling polymer known to inhibit protein adsorption and cell adhesion. Streptavidin, renowned for its strong and specific binding affinity for biotin, was chosen for its ability to anchor fibronectin onto the surface effectively. The objective of the experiment was to evaluate the cell adhesive properties of the modified surfaces. Three types of samples were prepared: surfaces coated with PLL-PEG-biotin (0.1 mg/mL), surfaces initially coated with PLL-PEG-biotin (0.1 mg/mL) followed by streptavidin-fibronectin treatment, and surfaces directly treated with fibronectin (5 μg/mL). HUVECs were seeded onto the surfaces, and after 24 hours, they were stained with DAPI and Phalloidin to visualize nuclei and cytoskeletal structures, respectively.
Results:
Surface Characterization:
Plasma treatment for 1 minute was shown to provide an effective PLL-g-PEG-Biotin coating due to an adequate number of negative hydroxyl groups on the surface. Longer plasma treatment did not show any improvement in the surface coating condition. Topography imaging with AFM revealed distinct surface profiles between the coated and non-coated surfaces (Figure 2A). The root mean square roughness (Rq) was measured as 1.325 nm for the non-coated surface and 4.094 nm for the coated surface, indicating the presence of PEG molecules on the surface. Fluorescent labeling of the biotinylated surface using Alexa Fluor™ 488 streptavidin exhibited a significantly higher intensity on the coated surface compared to the non-coated silicon nitride surface, consistent with AFM topography images of the surfaces (Figure 2B). Contact angle measurements demonstrated an improvement in surface hydrophilicity after coating, confirming the presence of hydrophilic PEG molecules (Figure 2C). SEM images of coated and non-coated membranes exhibited no significant pore clogging post-coating, with only 13% reduction in pore diameter, indicating minimal impact on overall porosity (Figure 2D). This can be attributed to the non-crosslinked configuration of the PLL-g-PEG-Biotin, as the polymer chains are not chemically bonded together to create a dense structure and clog the pores.

Thickness Measurement:
AFM imaging in a PBS environment captured a comprehensive 2D height image of the surface, revealing distinct features corresponding to the PEGylated spots and non-PEGylated regions. Analysis of the height profile along a designated white line within the image highlighted the height disparity between the PEGylated and non-PEGylated areas. This difference in height, measured at 21.95 nm, was interpreted as the thickness of the PLL-PEG-Biotin brushes on the surface (Figure 3B). Notably, the upper red line denoted the average peak height, while the lower red line represented the average valley depth, further delineating the surface topography.
Cell Adhesion:
The cell adhesion experiment revealed notable differences in cell behavior on various surface treatments. Initial observations depicted a non-adhesive PEGylated surface (A), where minimal cell attachment was observed. However, upon treatment with Streptavidin-fibronectin (B), a significant improvement in cell adhesion was evident, indicating the efficacy of the surface modification in promoting cell attachment. Furthermore, comparison between the PEG-fibronectinylated and fibronectinylated surfaces revealed distinct cell morphology differences, with HUVECs exhibiting a more rounded shape and reduced spreading area on the PEGylated surface. This observation suggests a softer surface characteristic on the PEGylated substrate, potentially influencing cell-substrate interactions.
Reference:
[1] Timpl, Rupert, and Judith C. Brown. “Supramolecular assembly of basement membranes.” Bioessays 18.2 (1996): 123-132.
[2] Palchesko, Rachelle N., James L. Funderburgh, and Adam W. Feinberg. “Engineered basement membranes for regenerating the corneal endothelium.” Advanced healthcare materials 5.22 (2016): 2942-2950.
[3] Halfter, Willi, et al. “New concepts in basement membrane biology.” The FEBS journal 282.23 (2015): 4466-4479.
[4] Van Helvert, Sjoerd, Cornelis Storm, and Peter Friedl. “Mechanoreciprocity in cell migration.” Nature cell biology 20.1 (2018): 8-20.
[5] Carter, Robert N., et al. “Ultrathin transparent membranes for cellular barrier and co-culture models.” Biofabrication 9.1 (2017): 015019.
[6] Youn, Jaeseung, et al. “Thin and stretchable extracellular matrix (ECM) membrane reinforced by nanofiber scaffolds for developing in vitro barrier models.” Biofabrication 14.2 (2022): 025010.
[7] Liliensiek, Sara J., Paul Nealey, and Christopher J. Murphy. “Characterization of endothelial basement membrane nanotopography in rhesus macaque as a guide for vessel tissue engineering.” Tissue Engineering Part A 15.9 (2009): 2643-2651.
[8] Lauridsen, Holly M., and Anjelica L. Gonzalez. “Biomimetic, ultrathin and elastic hydrogels regulate human neutrophil extravasation across endothelial-pericyte bilayers.” PLOS one 12.2 (2017): e0171386.
[9] Zahra Allahyari, Shayan Gholizadeh, Henry H. Chung, Luis F. Delgadillo, and Thomas R. Gaborski, ACS Biomaterials Science & Engineering 2020 6 (2), 959-968, DOI: 10.1021/acsbiomaterials.9b01584
[10] Schindelin, J., Arganda-Carreras, I., Frise, E., Kaynig, V., Longair, M., Pietzsch, T., … Cardona, A. (2012). Fiji: an open-source platform for biological-image analysis. Nature Methods, 9(7), 676–682. doi:10.1038/nmeth.2019