Update on Anodic Bonding at RIT
Yesterday Henry and I went over to RIT to perform anodic bonding and parylene deposition using the new membrane configuration of W619 (with the etched trenches for our capillary tubing). I just wanted to post a summary of the process that was done yesterday, as we encountered numerous problems, in the hopes that we can come up with a few possible solutions.
Anodic Bonding:
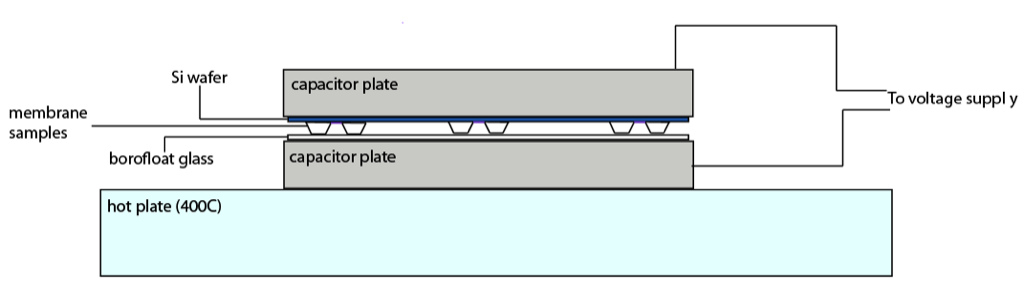
The diagram below shows a diagram of the “sandwich” created during the anodic bonding process…
A clean borofloat wafer is placed down on the base capacitor plate on top of which are placed our membrane samples (with membrane side facing up). In order to prevent damage to the membrane from the top capacitor plate a Si wafer must be placed on top of the membrane side of our sample chips.
Although Si wafers are “smooth” there is still damage caused by them being placed directly on the surface of our membrane. Out of the six intact samples that henry and I bonded yesterday only two survived the anodic bonding process intact. A thing to note is that W619 still has a protective oxide layer making them stronger then stripped membrane. This leads me to believe that if we were to try to bond stripped sample we would have an even lower yield.
To protect the membrane surface during the anodic bonding process there must be some sort of shim made, but at the same time it must be made from a conductive material to still allow bonding to occur. With this the issue of pressure distribution becomes important. If the shim causes an unequal distribution of the weight of the top capacitor plate then the sample chips can easily crack (something that was observed even without a shim during yesterdays bonding).
Parylene Deposition:
This process has turned out to be a little more troublesome than we initially anticipated. In order for the deposition to occur at uniform thickness the sample must be suspended in the deposition chamber. This means that we cannot place the membrane side of our chips on some sort of PDMS supports to protect them from being coated with parylene. Having our sample freely suspended means the “only” way to protect them would be to directly cover the membrane surface. Removing this protective barrier will most likely cause the membrane to break. Also, once the protective barrier is coated with parylene, removing it might cause the parylene deposited near the capillary tubing entrance to be pulled away.
After talking with Dean about yesterday’s deposition he feels that the etch we made in order to fit our capillary tubing is not small enough to be filled with parylene alone. He suggested first filling it with an epoxy or something similar.
Overall, I think that anodic bonding is a good idea and should be further pursued as long as we can find a way to protect our membranes. On the other hand I feel the parylene deposition is a very tedious and messy process that causes more issues than it helps to resolve, not to mention we do not have the necessary equipment on campus.
Some of these problems may not be an issue when making a two channel system with an internal protected membrane.
We always have the option of coupling from the top, through holes on glass or PDMS. I think this should at least be explored in parallel.