Smith Lab Diffusion Studies
Harold Smith at the Med Center proposed an experiment for Jess and I to try, in which we use our membranes to separate proteins onto a PES, PVDF (Polyvinylidene fluoride) or Nitrocellulose membrane, similar to a Western Blot of DNA microarray. The idea is to create a system to screen for drugs that break protein-protein complexes. Our pnc-Si membrane would hold back the protein-drug complex and allow the non-bound drug to pass through onto the other membrane for detection. Fluorescent dye will be used to park the drug for easy detection on the other membrane, and on the backside of our membranes.
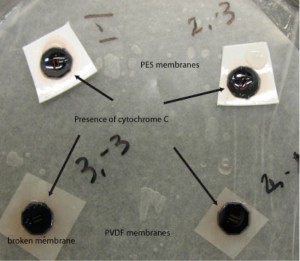
Yesterday I set up a diffusion system similar to our diffusion cell in which I placed 2 PVDF and 2 PES membranes in a humidity chamber. The PES membrane acted as our control since we already know that proteins stick to it. I washed the PES and PVDF membranes in PBS before placing them in the humidity chamber. Vacuum grease + microSi beads was applied to the edge of the membrane side of the chip. I applied 10uL of ddH2O to the membrane to prevent air pockets forming. I added 5uL cytochrome c to the wells and sealed the humidity chamber. After 19 hours it was apparent that the cyctochrome C passed through the membranes and stuck to the other membranes. The images are provided below:
The membranes used were from wafer 412, with pinholes. Position numbers (1,-1) two pinholes, (2,-1) two pinholes, (2,-3) eight pinholes and (3,-3) four pinholes. Only one membrane broke after the cytochrome C was added (3,-3).
Eventually we want to be able to create an array using our membranes in which we can pipette the protein + drug solutions onto the wells of our membranes and see if any of the drugs pass through the membrane. Ideally, we don’t want to see any of the drugs passing through because that proves that the drugs formed a complex with the proteins. The problem here is that the PVDF and nitrocellulose membranes allow the liquids to spread, which will not allow for easy detection of the drugs. Ideas to prevent spreading of the fluorescence including using PDMS to create a honeycomb type web to place the membranes on, or using electrodes to create an electric field. The electric field will allow the material to diffuse faster and will allow for easier set-up.





Can you remove the chips, rinse the membranes and check for color. I want to make sure the membranes are ‘stained’ by the protein.
I doubt spreading will be an issue unless the wells are very close together, provided the gap between our membranes and the nitrocellulose is small. Unlike PES, nitrocellulose is designed to readily adsorb any protein that would pass our membranes, before it diffuses away, assuming the drugs are proteins. PES does not adsorb much, as it is designed to be non-stick. Jess’ experiments are comparing all non-stick materials. Actually nitrocellulose would be an interesting positive control for adsorption and would likely show orders of magnitude more binding than PES, cellulose, pnc-Si, etc..
How do you make sure the drugs don’t diffuse through before they bind to the protein? Or are they mixed together prior to placement on the membrane?
The key to this idea is the detection of protein on the polymer membrane that would otherwise be part of a complex held behind our membrane. The drug that breaks up the complex can equilibrate across the membrane and this idea should still work, although loss of drug from the filtrate side would be informative I suppose.
The original description of the idea is here …
I rinsed the membranes and posted a picture. The image on the left was taken as soon as I removed the membrane and the one on the right shows the membranes post-rinse. It is harder to detect the color on the PVDF membrane.
Take one more picture when both membranes have completely dried out.
Hmmm. PVDF is a blotting membrane, right? Or is this one treated in some way? Seems like it should be much darker than the PES. I’d be curious how nitrocellulose compares..
Detecting a protein that breaks away from a complex makes sense, as long as the initial solution is purely complexed. Would the protein be detected with a labeled Ab? Seems like an ideal application for a label-free method.
Yes, an ideal application for a label-free detector!
The picture of the membranes farthest to the right was taken after the membranes were able to completely dry out. The picture taken right after rinsing and the one taken after drying are very similar