Discoloration Study with Bacterial Media
I performed a test study to see the effect of microbial media’s : LB and BHI Broth on the membrane to check for discoloration.
LB Broth : Lysogeny broth,Luria broth or Luria-Bertani broth. Obtained from Sigma and made by dissolving 20g in 1 l of D.H2O and autoclaving at 121C for 15 mins. LB is simple media consisting of tryptone, NaCl and yeast extract.
BHI broth or brain heart infusion broth also from Sigma made by dissolving 37g in 1l of D.H2O and autoclaving at 121C for 15 mins. BHI is primarily made up of heart and brain extract from cows so there is a degree of variability associated with the media.
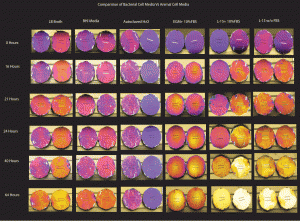
Wafer 670 was tested for discoloration. Samples used had torn membranes as these were the ones left over from the pressure tests. All wells contained 2 membranes ( duplicates)
Experiment was performed in a 6 well culture plate and incubated at 37C in the incubator. Control well contained autoclaved DD H2O (-ve control) . Positive control wells contained EGM+10% FBS, L-15 +10% FBS, L-15 w/o FBS.
Pictures were taken of the membranes before the experiment and then at regular time intervals: 16 hours, 21 hours, 24 hours, 40 Hours and 64 Hours.
Result: The negative control well did not show discoloration as expected even at 64 hours. The positive control wells showed discoloration in as little as 16 hours ( L-15 Media) and complete discoloration was seen in 64 hours ( L-15 Media). The BHI broth did not show any signs of discoloration even at 40 hours. However at 64 hours signs of discoloration began to set in. The LB broth samples showed slight discoloration at 21 hours not significantly changing even at 40 hours. 64 hours into the experiment, the membranes started discoloring. This is expected as the microbial media has a more regulated pH and does not contain the buffering agents present in the animal culture medias. pH of the LB broth:7.0+-0.2 BHI broth: 7.4 +-0.2 . L-15 Media : 7.4- 7.8 L-15 with glutamine: 7.3- 7.9.
Conclusion: Rather than the pH being the sole cause of discoloration, it might be the buffering effect of the agents in animal media combined with the specific components causing color change. I plan to see if media in the Sepcons affects the membrane and whether we can use centrifugal force to extract proteins from the media components.
Which wafers are these?
L15 is the positive control?
It’s depressing that L15 is now causing discoloration, although there is something different about these wafers. How did you measure the pH?
Also were any of these RTP treated?
What are the funny patterns, even on some of the 0 hours samples? I doubt they look like this on the wafer…
This was wafer 670 on which i had performed burst pressure. These were just lying around and we got new bacterial media so i did a pilot study to test the discoloration. L-15 i used as a control not a positive control. The EGM was the positive control but since there were 2 extra wells in the plate i decided to add L-15 to those and perform the experiment. I didnt measure the pH on these as this was just a starting experiment. The funny patterns could be since that these were used membranes.
670 may have been an unfortunate choice. We really don’t know if ‘new’ material is less chemically stable than ‘less pure’ material that we made a short while ago, only that 670 is new and seems to be more sensitive than any other wafer we produced.