Discoloration – Sepcon format and stresses
We’ve been wondering about the effects of membrane stresses on discoloration rates. This was spurred by Vlassak’s visit and his ideas about stress and membrane stability.
In this experiment, I used w501, no RTP, in the oven in RPMI+10%FBS. All of these samples were in the Sepcon format: chip sealed with a fat O-ring, chip sealed with the thin O-ring, chip sealed only with vacuum grease or chip with nail polish-covered membrane windows. The hypothesis was that the greased chip membranes would be under the least stress since there was no clamping. The fat O-ring sample might have less stress since the fat O-ring could potentilly absorb more stress than the thin O-ring.
The thin O-ring is what Anant and I have normally worked with – it’s not suprising that this didn’t discolor in 8 hours. The greased sample is totally discolored, though!? The fat O-ring sample is disoclored, as well (remember that the pnc-Si area exposed ot media is much lower in fat O-ring samples). Could stress actually be helping to slow down discoloration? If discoloration is caused by molecular attack at the pnc-Si/underlying oxide layer interface and then propagates by delamination, then stress might help to slow this down.
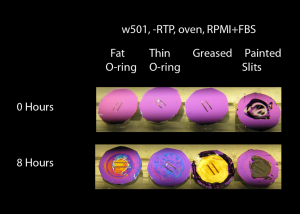
Since the greased sample in Sepcon discolored faster than the ones with o-rings, there is a possibility that the o-ring absorbs/adsorbs components from media and prevents the discoloration. We have also seen this in one of Barrett’s experiments where different Sepcon components were taken and it was observed that the sample alongwith just o-ring discolored slowest. Right?
One more thing. Why would the fat o-ring sample have lower stress? I was thinking the other way. (Jim also mentioned it in one of his comments)
I had a brief chat with microelectronic engineers at MIT last week- and they confirm that stress can change the way in which silicon is chemically attacked (they were not thinking of biological fluids, more like known etchants etc, but I think the concepts should be connected- after all, it’s all about chemical reactions!)
I still don’t like the stress theory, because we are talking about relatively tiny extrinsic stresses caused by an oring. Intrinsic stresses are much higher and are know to affect chemical processes.
Another possibility that I have been thinking about is whether gas generated by the dissolution reaction is passivating the surface. I beleive this process produces H2 gas that could coat the surface with microbubbles. I’m not sure how such bubbles would behave in the various experiments that have been performed, but it may be worth thinking about.
I sometimes notice bubbles during discoloration, more so when the discoloration is fast. But how would this explain the Sepcon vs. flat results?
I was thinking that bubbles should be considered for geometric factors. Samples facing down may gather more bubbles and confined geometries may slow the dissolution and transport of the gas away. Just a thought…