Discoloration Array with NaOH
All of this talk about Zeta Potentials, electro-osmosis and electric double layers got me thinking about these phenomenon in relation to discoloration. We’ve known that salts accelerate discoloration, and we know that bicarbonate solutions accelerate discoloration as well. I explained the bicarbonate issue based on the equilibrium between bicarbonate ions and hydroxyl ions (pH). We never really investigated what salts were doing to cause discoloration…..
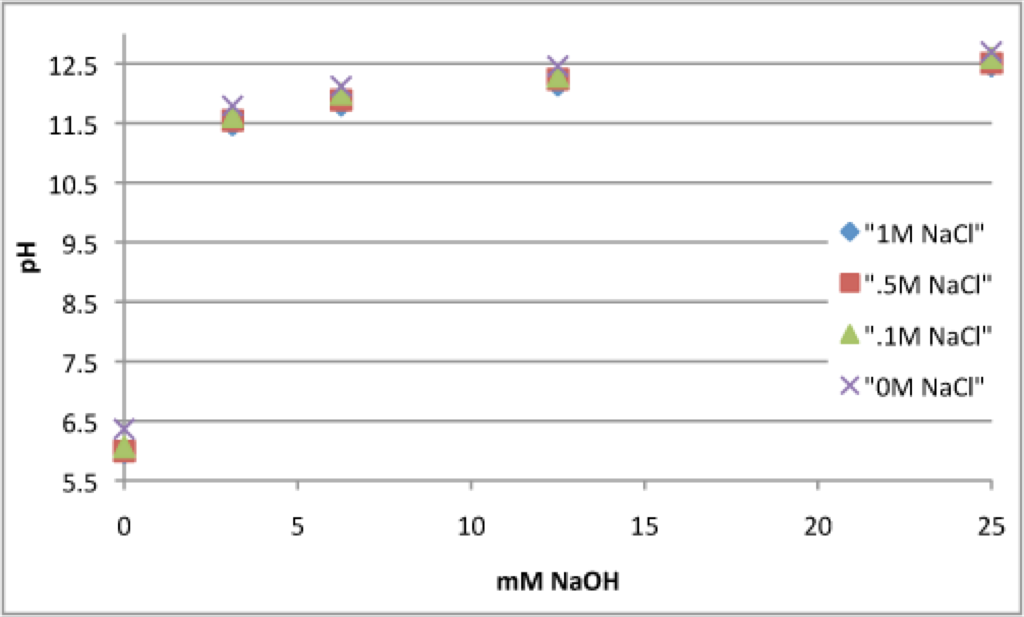
Last week I read a silicon dissolution paper and got to thinking about double layers and discoloration. Jess presented some math that showed that higher salt solutions decrease the Debye length. Do smaller Debye lengths permit easier access to the silicon surface for OH- ions to cause discoloration? This has been hard to prove based on our weird discoloration arrays (AAA: here, BJN: here, here). I decided to make the system simpler, so I looked at NaCl and NaOH (instead of NaHCO3):
pH of these solutions at time = 0 minutes. They’re basically the same across different salt concentrations.
The array looks exactly as we’ve been hoping for. Higher salts accelerate discoloration at a given pH (OH- concentration) and higher pH (higher OH- concentration) and a given salt concentration accelerates discoloration. Since OH- ions are the major species responsible for discoloration, increasing the pH will accelerate discoloration. With higher salt concentrations, the Debye length is lower and I hypothesize that this allows more OH- ions to interact with (and discolor) the silicon surface.
Why doesn’t this array work with NaHCO3? NaHCO3 dissociates into Na+, HCO3-, H2CO3 and OH-. Can the array be explained like this?: at a given salt concentration, low bicarbonate levels cause discoloration because of the addition of OH- ions, at “medium” concentrations of NaHCO3, HCO3- ions push OH- ions away from the surface and slow discoloration and then at high bicarbonate levels, the pH is high enough to cause discoloration despite the presence of HCO3- ions?

This is good news.
We will need a complex model to answer your question about bi-carb: multi-species debye layers and multi-species chemistry. Complex, but doable.
I’m not sure if the debye layer theory will fly in a publication, because although the layer is thinner, the potential barrier that must be overcome is the same height, right? Since the ion concentration is higher, this potential is screened over a shorter distance, however the reality is that it makes the hill steeper that you have to climb to reach the surface. I would favor an explanation where the higher surface concentration of ions from solution catalyzes some chemistry at the surface. It would be nice if we could identify someone on campus who could shed some light on this. It’s more complicated than just the DLVO theory, since there is a reaction going on simultaneously, and therefore the surface is in a dynamic state…
If the same energy input can bring a molecule within reach of a reaction site in the presence of salts, then the reaction should happen with greater frequency. From my readings, salts are known to impact the ‘activity’ of molecules in reactions because of their ability to shield charges. The help of a solution chemist would be nice, particularly for the bicarb effect. But I don’t think the salt effect is terribly controversial.
If there is another effect involved, such as hydrophobic interactions or large entropy changes, I can see where charge screening can help two parties get-together. However in this very simple case of hydroxide attack of a surface w/ and w/o salts, I’m less convinced. I don’t see how a more “dense” but thinner debye layer is any better than a thicker less dense layer at allowing hydroxide ions to reach the surface. At the end of the day, I suspect that there is some electrochemical effect that helps drive the dissolution, beyond just the debye layer thickness.
We should be able to calculate the potential at the Debye length for these different solutions. I think this might help us think more clearly about thin/dense vs. thick/dilute Debye layers. I’m also going to try this array with a large ion (instead of bicarb) to see if the trend is the same.
Hi, this is my first try at this blog site, so forgive any screwups. My grad student, Paul Black, and I have been working with membranes kindly provided to us via Jess and Jim. Our application involves DNA filtration, something Paul and I will expand on latter. We have been climbing the learning curve on how to minimize breakage, maximize reuse of the same membranes, and avoid things like base catalyzed destruction. Without a doubt, we are repeating experiments you have done earlier and better. One of them being the pH effect.
With that intro, I wanted to comment on a way to test the concept of increasing OH- encounter by increasing the anion concentration. If this is due to screening vs. some catalytic impact of the anion, it seems this might be tested by changing anion size. And from your last post, this already occurred to you. But you might go down in anion size as well as up. You might test the extremes: LiCl and and CsCl. These should influence the Debye length and the entropy of the system in different ways.
Bill Bernhard
Thanks for the suggestions. We don’t have those 2 salts in the lab now, but we should be able to acquire them. I was going to use sodium fluorescein and think about the dissociated fluorescein(-) as a huge anion. Perhaps a divalent salt would be illustrative as well.
I just checked, we have LiCl (500 g bottle) and CsCl (100 g bottle). You are welcome to borrow the bottles or scoop some out. I am in B-6835 of the med center. I think Cs would be a bit easier to understand than fluorescein. Anyway, it is here if, or when, you might want to try it.
Since we have just started to play with NaCl concentration and see a really nice effect on DNA filtration, we may look at some other salts as well. Paul and I will put together a report on our findings, so you can see what we have been up to. Coming soon.
Check out:
http://www.freepatentsonline.com/4781853.html
Apparently there is a patent titled “Method of enhancing silicon etching capability of alkali hydroxide through the addition of positive valence”. The claim is that the extra positive ions facilitate electron extraction from the silicon lattice, making it more susceptible to hydroxyl attack. It’s not a peer-reviewed journal, but may have some merit.
I suppose this is related to the concept that Jess mentioned in her talk about how adding salt will lower the Zeta potential due to the adsorption of positive ions to the solid surface. Those adsorbed ions will perturb the electrons in the surface bonds, potentially making it easier to pick off a Si atom.
Another theory that Ben Miller mentioned was the enhanced solubility of the silicate reaction product in the stronger ionic solution. Obviously, this would drive the reaction toward dissolution. I could buy this too.