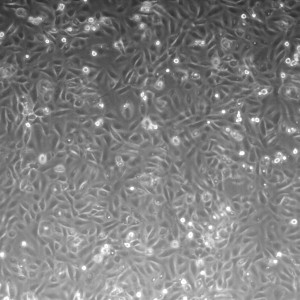
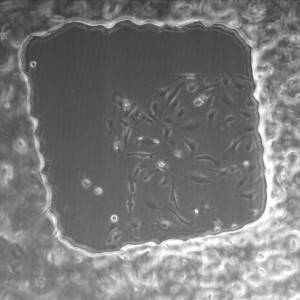
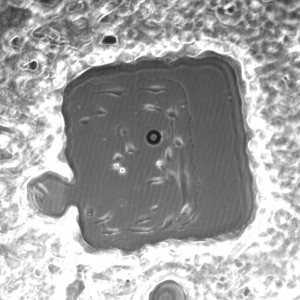
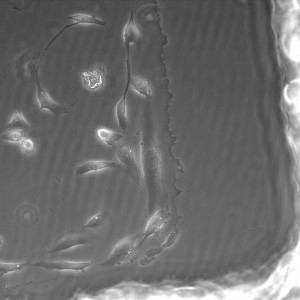
20x shown below.
This data prompted us to do an experiment where the PEG was diluted before crosslinking. We also decided to test different concentrations of photo initiator.
Varying concentrations of photo-initiator (3%, 1.5%, and 0.3%) were tested with 1:1 PEG dilutions with DI or PBS.
Exposure times:
3% – 500ms
1.5% – 2000ms
0.3% – 20000ms
Using DAPI filter cube without an objective.
0.3% did not effectively crosslink with either DI or PBS in dilution. Previous efforts to crosslink 0.3% photo-initiator in 100% PEG were successful, but I am assuming there is just not enough PEG or it to far spread apart in the dilution to effectively crosslink, even with a 20 second exposure time.
The microarrays were crosslinked on the microscope and washed once in their respective solutions. For the microarrays diluted in water, there was a second wash done in PBS to observe any effects of contraction.
All images were taken at 4x.
1.5% initiator in PEG+DI – first wash with DI (Maybe this was just a badly TPM’d slide?)
1.5% initiator in PEG+DI – second wash with PBS
3% initiator in PEG+DI – first wash in DI
3% initiator in PEG+DI – second wash in PBS
1.5% initiator in PEG+PBS
3% initiator in PEG+PBS
12 Days Later: Microarrays were allowed to sit in either DI or PBS for the length of time.
3% initiator in PEG+DI
1.5% initiator in PEG+PBS
3% initiator in PEG+PBS
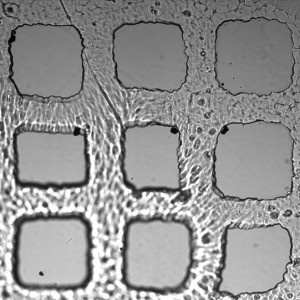
PBS looks like a better choice to keep the microarrays attached to the coverslips. 1.5% photoinitiator gives the most uniform features with the crosslinking conditions I being used.