Creation and Characterization of Silver Chloride Electrodes
Silver chloride electrodes are useful for microfluidic devices since they can generate current below one volt. This allows them to function without fear of electrolysis confounding experimental results by way of gaseous clogging. Several different methods for coating silver mesh were tested to determine which would be ideal for future experimentation. These methods were as follows: (1) coat the silver mesh in a chloride ink, (2) soak the silver mesh in bleach for 40 minutes, (3) electroplate chloride onto the mesh.
The advantages and drawbacks of each method were as follows:
Chloride Ink |
Bleach Soak |
Electroplating |
| +Ease | +Ease | -Ease |
| +Longevity | -Longevity | +Longevity |
| -Cost | +Cost | +Cost |
| -Purity | -Quality | +Quality |
To evaluate the performance of various coating methods, electrodes with an area of roughly 38 mm^2 were placed on opposite sides of a 50 mL beaker filled with 0.025 M KCl solution. A constant voltage was then applied and the resulting current was measured over time. The chloride ink performed as follows:
While this represents relatively good longevity, it also turned the solution a silvery color and caused strange discoloration patterns on the electrodes:
This raised some concerns about the purity of the coating, so other methods were investigated.
The next method investigated was simply to place silver mesh into bleach and let it soak for a while. This was tested with 40 minute soaks both at room temperature and at 50 C. These electrodes died very quickly:
Here “post-prime” indicates performance after the system had already been run in one direction (so all of the chloride from preparing the two electrodes had moved onto one side). The performance reliably improved post priming. This seems to indicate that additional coating would improve performance, but soaking the electrodes for several hours did not significantly enhance their longevity.
In addition to the pitiable deposition volume of the beach soak, the coating quality was also sub-optimal. It tended to lead to large masses of junk breaking off of electrodes which could easily clog devices:
Pictured above: Junk that could clog devices.
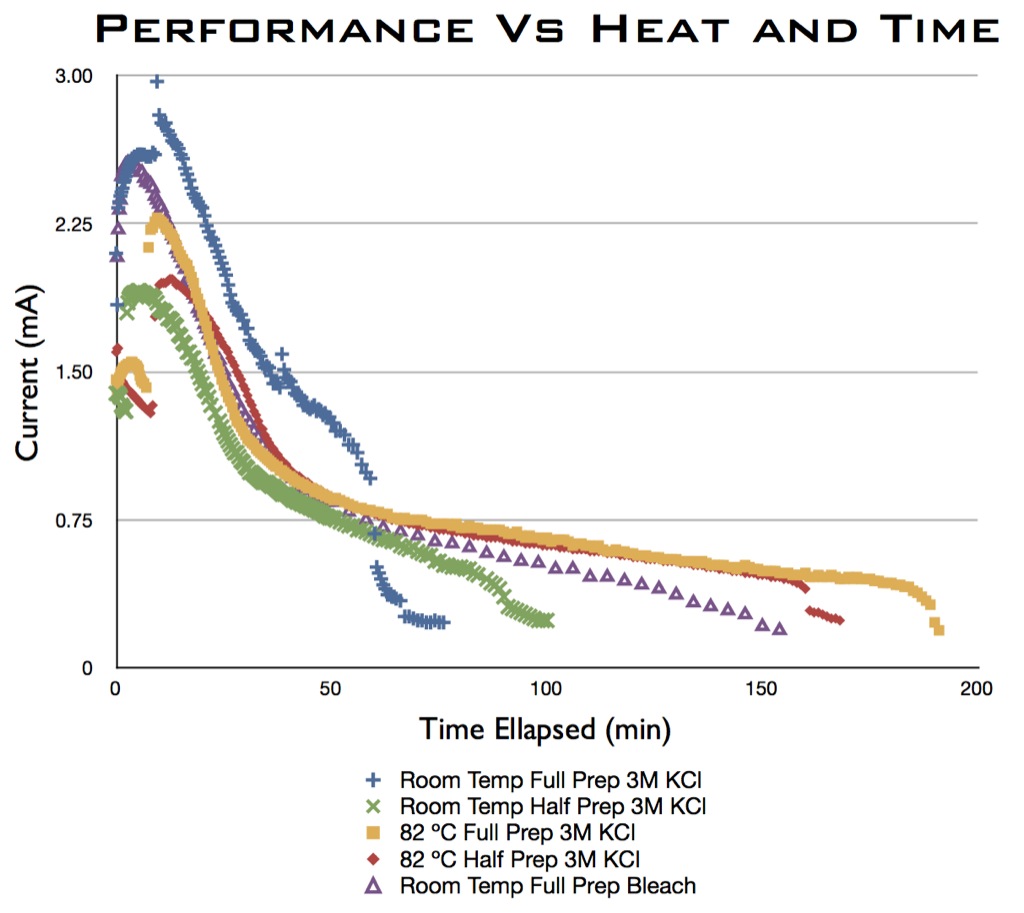
The final method investigated involved electroplating chloride onto the silver mesh electrodes. This was tested both at room temperature and at 82 C for various durations, and with both 3M KCl and bleach as the electroplating solution. The electrode performances are summarized as follows:
Hot KCl preparation is the clear winner as far as longevity is concerned. It also produces a higher quality (more uniform) coating than the bleach methods:
The only downside of the hot KCl preparation is that it results in crystal formation on the mesh. This can be dissolved off with a 12 minute DI water bath, though longevity suffers slightly (they still last more than 2 hours though).
Of the methods investigated, it seems heated electroplating in 3M KCl solution is ideal. The procedure for this process is as follows:
| (1) Heat a 3M KCl Solution for 30 sec in Microwave |
| (2) Transfer to hotplate and bring to 82 C |
| (3) Submerge electrodes and apply +9 volts DC for 8 min |
| (4) Apply -9 volts for 8 min |
| (5) Apply +9 volts for 5 min |
| (6) Apply -9 volts for 5 min |
| (7) Remove electrodes and soak in DI for 12 min |
The electrodes should then be ready for use in microfluidic devices. Alternating the voltage several times throughout the coating process makes the electrodes coat better, though the exact reason for this is unknown. One explanation is that the reversal pulls off the surface layer of silver chloride allowing more of the mesh to be exposed for a deeper coating. If the electrodes are being used in a closed system they can simply be run backwards after depletion to recharge.
Edit:
I recently tried these experiments again with two different procedures. Both involved trading the 3M KCl for 0.1 M HCl.
Procedure 1: 26 minutes at 2 V alternating polarity every 5 seconds. These electrodes died after less than 1 minute when placed in 0.025M KCl for testing.
Procedure 2: 7 point electroplating procedure as outlined above but without heating or DI soaking (since soaking reduces the longevity of these coatings). These electrodes lasted 42 minutes on average. Below is the precise performance of the electrodes on the same scale as the graph of performances above. Not only did the electrodes perform worse, but the coating process seemed to eat away at them significantly. Unless there is some nuance of using HCl which I am unaware of, I would recommend sticking with the 7-point procedure outlined above with KCl for electrode creation.




Excellent post! I tried my hand at electroplating last week, and your protocol looks substantially better than the one I was using. Is there any chance you could put up the best performances of bleach, ink, and electroplating on the same chart? I’m also curious what your setup looks like.
For the record, here’s the only previous electrode recipe on the blog: https://trace-bmps.org/data/2008/12/30/agagcl-electrodes/
and here’s the recipe I was working off of:
http://www.warneronline.com/Documents/uploader/Chloriding%20Ag-AgCl%20electrodes%20%20(2004.02.02).pdf
I have not tested the ink system to failure at 1 volt, so I cannot make that chart yet. I could do that once I get my current EO project working. Integrating under the curves I do have (to get total charge moved) indicates that electroplating gives a longer half life than ink.
The setup is just a 50 mL beaker sitting on a hotplate with electrodes clamped at each end.
Thanks for this very helpful post. Sadly even the ‘ideal’ case gives time dependent performance.
Jirachai had also tested this during EO and found stable currents. I believe these were with AgCl electrodes. I’m not sure how long Jirachai measured current for but his work suggests more predictability. Here is a link to an old presentation Jirachai made at NRG.
I don’t seem to have access to that link, but the chloride ink I was using was borrowed from Jirachai and clearly exhibited time dependent behavior. I believe for his applications he was only running the electrodes for a short time. He told Bob recently that running electricity for extended durations creates ion gradients in the solution which he wanted to avoid as a potential confounding variable.
Even though the Ag/AgCl electrodes are known to be the non-polarizable electrodes, it is true only for the small current density. The fact is the electric double layer is formed on the surface of electrodes and behaves like a capacitor. When operating in DC mode, the measured current exhibits a transient behavior. It, however, depends on the current density on the electrode surface. If the electrode surface is large (capacitance is high) and the current is low (Faradaic reaction is low or resistance is high), the RC time constant will be large. As a result, the decay of current is slow. If the measurement time window is small, the fluctuation will be very small. In case of 100mM NaCl with 500mV of applied voltage and the electrode spacing from the pnc-Si membrane of about 2 mm, the measured current was about 275 uA with 5 uA fluctuation over 20 seconds measurement time window. The measurement time window was limited by the velocity of menisci as they must remain in view under the microscope.
Try again. I had to throw a switch to give you access to the folder.
From my experience with AgCl microelectrodes for electrophysiology it can be advantageous to slightly acidify the electroplating solution (0.1M HCl) – the conductivity of the 3M KCl will cause you issues as it will encourage large current flows between the electrodes.
At 9V in such a conductive solution you will be running things very “hot” indeed!
I would suggest you try a rather dilute electrolyte such as 0.1M HCl in dH2O with no further salts present.
In addition, to avoid the polarization issue as highlighted above it is also good to flip the polarity regularly – every few seconds ideally. We do this in the lab using a double-poled switch to connect the two electrodes to -ve and +ve in opposition which reverses when the switch is flipped.
Finally i think you’ll find that 9V is excessive, we usually use only 1.5-2V in the lab, since that is more appropriate to the electrochemistry of the AgCl half-cell. the layer you deposit will look black initially, fading up through dark grey to a matt mid grey color.
A final note – AgCl electrodes are also light sensitive, and will darken in color if you expose them to too much light.
I know this got kind of old, but I just tried the following coat procedure:
26 mins in 0.1 M HCl (same time as my procedure above) at 2V with 5 second polarity flips. I then put the electrodes into 0.025 M KCl to compare their longevity with those outlined above. The electrodes failed after less than 1 minute. During the coating process a lot of silvery looking stuff sank off of the electrodes. How long do you wait before changing the direction of current?