Incorporating a ECM hydrogel into an in vitro microvasculature system – Part III
During the last weeks of my rotation, Tejas and I attempted to study neutrophil migration in the following setup, using fMLP as a chemoattractant:
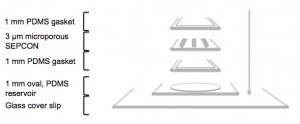
In order to do this, we permanently bonded a 1 mm PDMS gasket to the bottom and top sides of a 3 micron microporous SEPCON and filled the back side (which is facing down in the diagram shown above) with 15.6 mg/ml Geltrex. The gel was thermally polymerized at 37 ºC for 40 minutes. Since the working volume is, in this case, about 20 μl, we tried to prevent evaporation by enclosing the gasket/chip/gasket sandwich in a 35 x 10 mm petri dish sealed with Parafilm, along with a wet Kimwipe to keep the environment relatively humid.

Then, we added 30 μl of 100 nM fMLP or HBSS++ (for the control) to an 1 mm oval, PDMS reservoir permanently bonded to a 18 x 40 mm glass cover slip and laid the sandwiched chip on top of it for 1.5 hours to allow the chemoattractant to diffuse into the gel. After that time, we filled the top side of the gasket/chip/gasket sandwich with isolated neutrophils (hPMNs) and incubated the device at 37 ºC in the same enclosed arrangement described above. This is how the final ensemble looks like:
1.5 hours and 3 hours later, we performed manual Z-scans of the back of the chip .
We repeated this experiment a couple of times under different conditions:
1) Using chips without membranes
At first, we were just interested in assessing whether human neutrophils were able to crawl through 15.6 mg/ml Geltrex when in the presence of a chemoattractant (without adding the complication of having a membrane, at least for this first test). Z-scans of the back of two membrane-less chips under control conditions or in the presence of fMLP, respectively, are shown bellow.
Control (for this experiment, we filled the oval reservoir with 1x PBS instead of HBSS++):
fMLP (100 nM):
The first image, in both cases, was taken in a Z-plane that is near the level of the chip windows. Later Z-planes were taken as we went through the gel towards to back of the chip and then back again towards the front side. In the control experiment, most of the neutrophils were located near the window level of the chip. The neutrophils, in the arrangement containing fMLP, were observed towards a middle slice within the gel in the back of the chip.
2) Using chips with 3 micron microporous membranes (wafer #4400)
Control:
fMLP (100 nM):
Both Z-scans, in this case, start, approximately, at the back of the chip, close to the glass cover slip surface, and get progressively closer to the plane of the membrane. Interestingly, even if we observed a rather distinctive number of neutrophils below the membrane in the sample containing fMLP, we also found neutrophils at various Z-planes in the control. We repeated this experiment in order to asses the validity of this result.
3) Using chips with 3 micron microporous membranes (wafer #4400) and BSA coating
For this last attempt, we pre-coated the flat side of the chips with 1 mg/ml BSA for 1 hour at room temperature. Then, the BSA was carefully removed and the top of the chips were washed twice with 1x PBS, before adding hPMNs. Here, we show manual Z-scans of the back of the chip taken 1.5 hours and 3 hours after introducing neutrophils into the system.
1.5 hours,
Control:
fMLP (100 nm):
3 hours,
Control:
fMLP (100 nM):
Just as in the previous experiment, we observed neutrophils crawling through the gel in back of the chip when in the presence of fMLP, but also under control conditions. We noticed, however, that their number and relative localization were different in each sample. While we observed clumps of neutrophils in Z-planes near the glass cover slip when the chemoattractant was present, we saw fewer neutrophils (and only in planes closer to the membrane) in the control. It is possible that adding HBSS++ to the oval reservoir in the control, might have contributed to this effect. Noticeably, we didn’t see any appreciable neutrophil migration in the control sample of the membrane-less experiment when we used 1x PBS.