NP and IgG separations using wafer 1142 (14%, 36nm)
We used chips from the latest 5 slot SepCon NPN wafer 1142 (14%, 36nm) to test our ability to separate 20 nm “blue fluorescent” polystyrene NPs from Invitrogen from IgG.
We used a concentration of 10^14 particles/ml NPs and 1 mg/ml IgG. (This is a mixture of fluorescent and unlabeled IgG)
We compared forward vs reverse centrifugation at 3000 rpm in angled rotor centrifuge. We prewet the top and bottom with 10 ul of the buffer. (1xpbs, 0.1% Tween 20) We loaded 300 ul in the top (or bottom) and spun them for 60 minutes. The forward sample had 69 ul of filtrate. The reverse had up to 120 ul of filtrate, but I measure the loss in mass from bottom centrifuge tube and this process is prone to error.
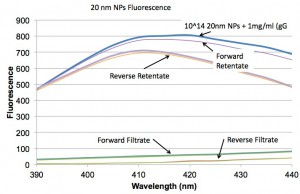
In this first plot we look at the fluorescence from 20 nm NPs and we see that both forward and reverse centrifugation with these wafers is doing a fairly thorough job of removing the NPs. (I think the rententate peak is lower than the original solution because it is slightly diluted by the prewetting, but I haven’t proven it.)
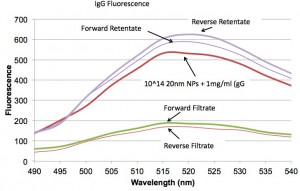
Next we see that roughly the same amount of IgG is getting through the membranes in both orientations.
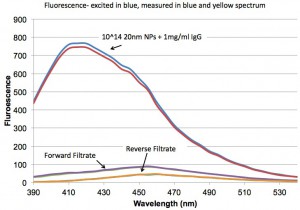
This final plot shows the a larger range of wavelengths scanned after exciting in the UV. We see a peak at ~450 nm that I can’t explain yet.
We should still run an IgG only filtration to make sure that it would flow unimpeded without the NPs in the solution. We are also pretty confident that IgG is binding to the NPs, so we should come up with a way to test if our reduced IgG concentration in the filtrate is at least in part a product of this binding.
Interesting. How do the filtrate volumes compare to other runs?
We don’t have an exact comparison, but looking back through similar tests I would say we are getting filtrate volumes within 10 ul or so for the forward orientation.