Vapor Condensation Inside a Nanocavity
My recent work has focused on wetting the inner portion of an attocavity with isobaric cooling to create condensation. We are trying to attack the problem of wetting through the nanomembrane without breaking the membrane. A perfectly dry membrane will always rupture before the critical radius for wetting the backside of the membrane is achieved. To counter this problem, we are trying to use residual water vapor to get more water on the backside of the membrane, allowing the critical radius to be achieved with less pressure from the frontside.
Assuming the following structure, how much water condenses on the inner surface, after a water bubble (sink) is placed on top of the outer surface, at standard room temperature and pressure, and cooled to 0 degree C (but not frozen)?, How much water vapor is necessary to break surface tension and allow water to infiltrate the inner cavity?
Constants
, Body Temperature, 308 K
, Room Temperature = 298 K
, Chill Temperature = 0 C = 273 K
, Standard Pressure = 1 atm = 760 T0rr
, Ideal Gas Constant = .082057 (L*atm)/(mol*K)
, Avogadro’s Constant = 6.023e23 #/mole
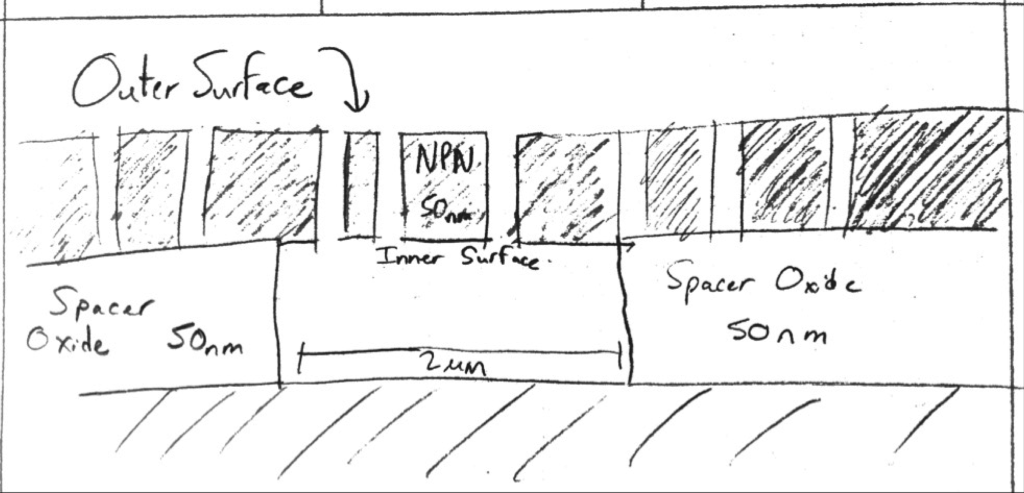
, Height of Cavity = 200 nm
, Diameter of Cavity = 2 microns
- Molar weight of water 18 g/mol
- 1 mmHg = 1 Torr
Calculated Constants
= Volume of Cavity (cylindrical) = 628e-18 L
, Vapor Pressure of Water (308 K) = .0555 atm
, Vapor Pressure of Water (298 K) = 23.6 Torr = 3.12e-2 atm
, Vapor Pressure of Water (273 K) = 6.44e-3 atm
, Avogadro’s Constant = 6.023e23 #/mole
This is a standard combined gas law problem, in the space of really tiny volumes
Assumption: The cavity is completely dry, with the bubble on top of the outer surface
I’m using the vapor pressure approximation for water, accurate to ~1%

How many moles of water will exist in the cavity in vapor at room temperature?
which is
How many moles of water will exist in the cavity after the the chamber is cooled to 0 C?
Assumption: the water molecules condense evenly everywhere in the cavity, then form tiny dew droplets
This is a fraction of the water that was originally present.
Therefore, 79.4% of the water condenses on the whole cavity surface.
How much water vapor will condense on the inner nanomembrane surface?

Will these molecules form dew droplets on the inner cavity?
Based on my experiments, we can see droplets shrinking away on the outer surface by evaporation around the ~5-10 micron size, microscopically looking like a fine mist.
Conjecture: the droplets on the inner surface must be significantly smaller if they are to exist within the cavity. An equivalent volume for a water droplet that would fill the 628 attoliter cavity would have a radius of 5.31 microns.
What radius of bubble forms from a nanopore, with normal hydrostatic pressure? Assumption: hydrostatic pressure is from a 10 um diameter water bolus, averaged out over the inner membrane cavity area
The amount of water in that spherical droplet is 523 attoliters, at a density of 1 g/mL, so the weight of the water is 523e-18 kilograms, spread out over an area of 3.14e-12 m^2, resulting in a hydrostatic pressure of 1.67e-6 N/m^2. Therefore the radius is
The water extending into the inner cavity is very flat on the surface.
Can the dew droplets on the inner surface merge with the bubble that forms from a nanopore?
Assumption: There’s enough energy for droplets to break surface tension.
How much water is necessary to get past the Laplace pressure (the radius decreases as the pressure increases)? Or alternatively, what critical water vapor density is needed to allow normal flow through the active area?
We know breath from Karl has enough residual water to get the desired effect, on a SEPCON chip under a mild amount of pressure (~5 psi).
Assumption: The water produced by breath in the lungs is 95% relative humidity at 35 C, producing a density of 3.767e-2 kg/m^3 of water vapor (http://planetcalc.com/2167/). The breath passes a volume of 50 mL over the entire flat side of the chip (29.16 mm^2) at room temperature (25 C).
Locally, we can do the same calculations as before and figure out the density of water condensing on the surface of the SEPCON chip.
so 42% of the water vapor has left and condensed on the the surface, and from the density of water vapor in the breath,
Assumption: The water vapor condenses evenly on the surface.
Remarks
The molecular areal density from Karl’s breath is over ~180 million times greater than the density in the nanocavity (4.995e4 molecules/um^2). We know Karl’s value works, but we don’t know if it is a critical point. Our experimental evidence suggests that the water is moving into the nanocavity, but we still haven’t confirmed it 100%. It may also be a question of scale, where we can clearly see a wetted surface in the microholes, but would be less obvious in the macroscopic wold. Would less humid gas work too? We could design a microchannel such that we could vary the absolute humidity in the bottom channel and try and get water to move from the top channel.
I’m leaning towards a dynamic transient model of wetting the nanocavity, where a critical droplet forms inside and draws the rest of the water in; the whole cavity is not wet at once, and the gas can escape through other pores.