BMES 2019 Recap: Organ-on-a-chip models with emphasis on modeling blood-brain barrier
There were lots of interesting talks and posters on organ-on-a-chip models with emphasis on modeling blood-brain barrier. I have selected four of them as a summary.
3D model of diffuse intrinsic pontine glioma growth, migration and blood-brain barrier interactions
Alanna M. Farrell, Raleigh M. Linville, Gabrielle N. Grifno, Peter C. Searson
Johns Hopkins University, Department of Biomedical Engineering, Baltimore MD
Related paper published a month ago: https://www.nature.com/articles/s41598-019-50193-1
This group generate tissue-engineered 3D models of the human BBB and tried to address 4 issues: (1) cell source – they reproduced a recently reported directed differentiation of derived human brain microvascular endothelial cells using fluorescently-tagged hiPSCs which enable live-cell imaging, (2) barrier function, (3) cryopreservation vs. fresh BMSCs (4) the role of matrix stiffness in BBB structure and function – the solute permeability of microvessels remains low across matrix stiffness ranging from 0.3–3.3 kPa, while the structural stability and dilation response were stiffness-dependent.
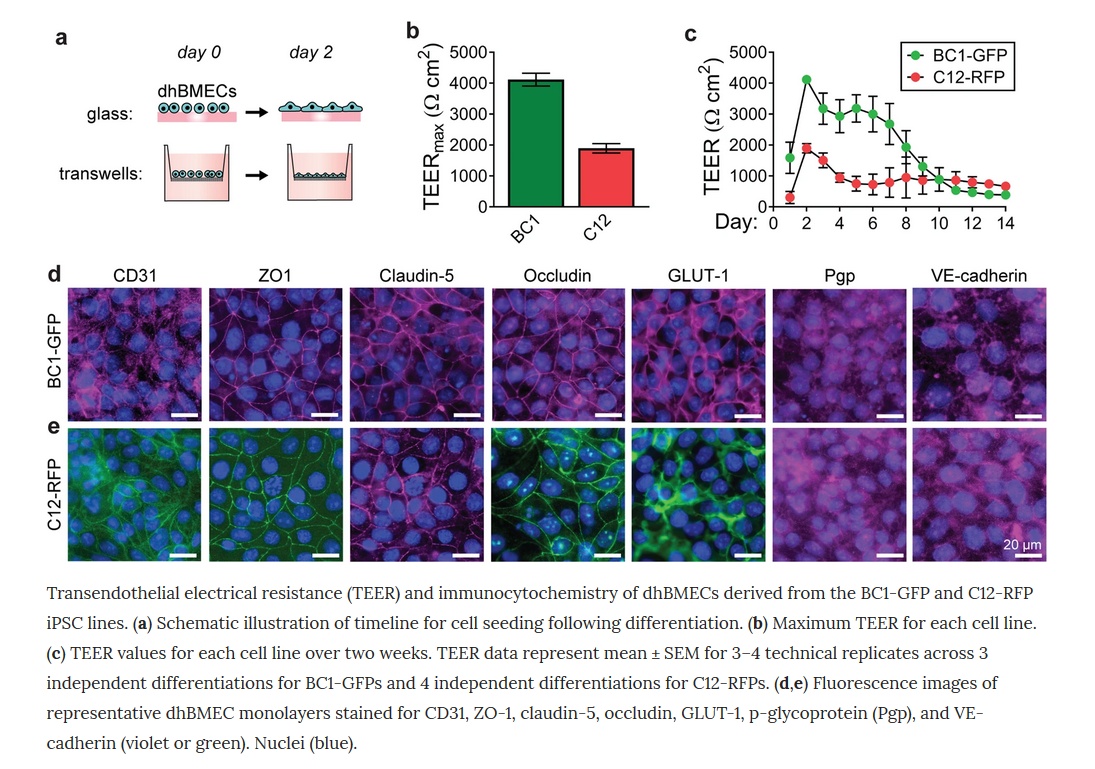
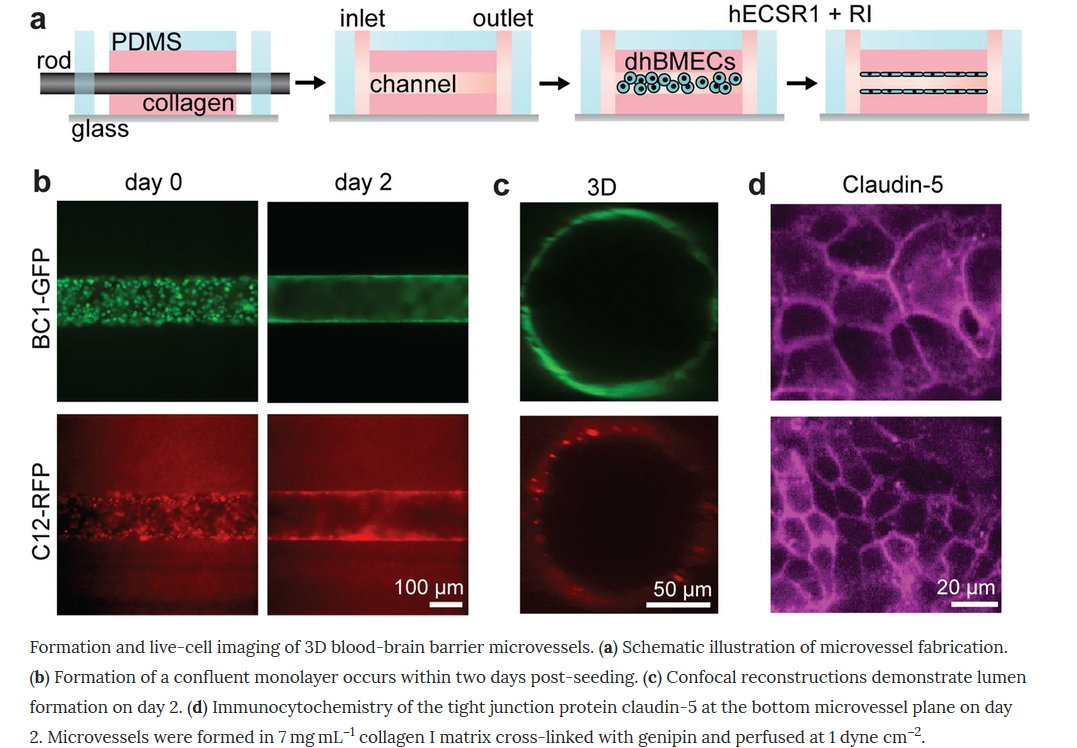
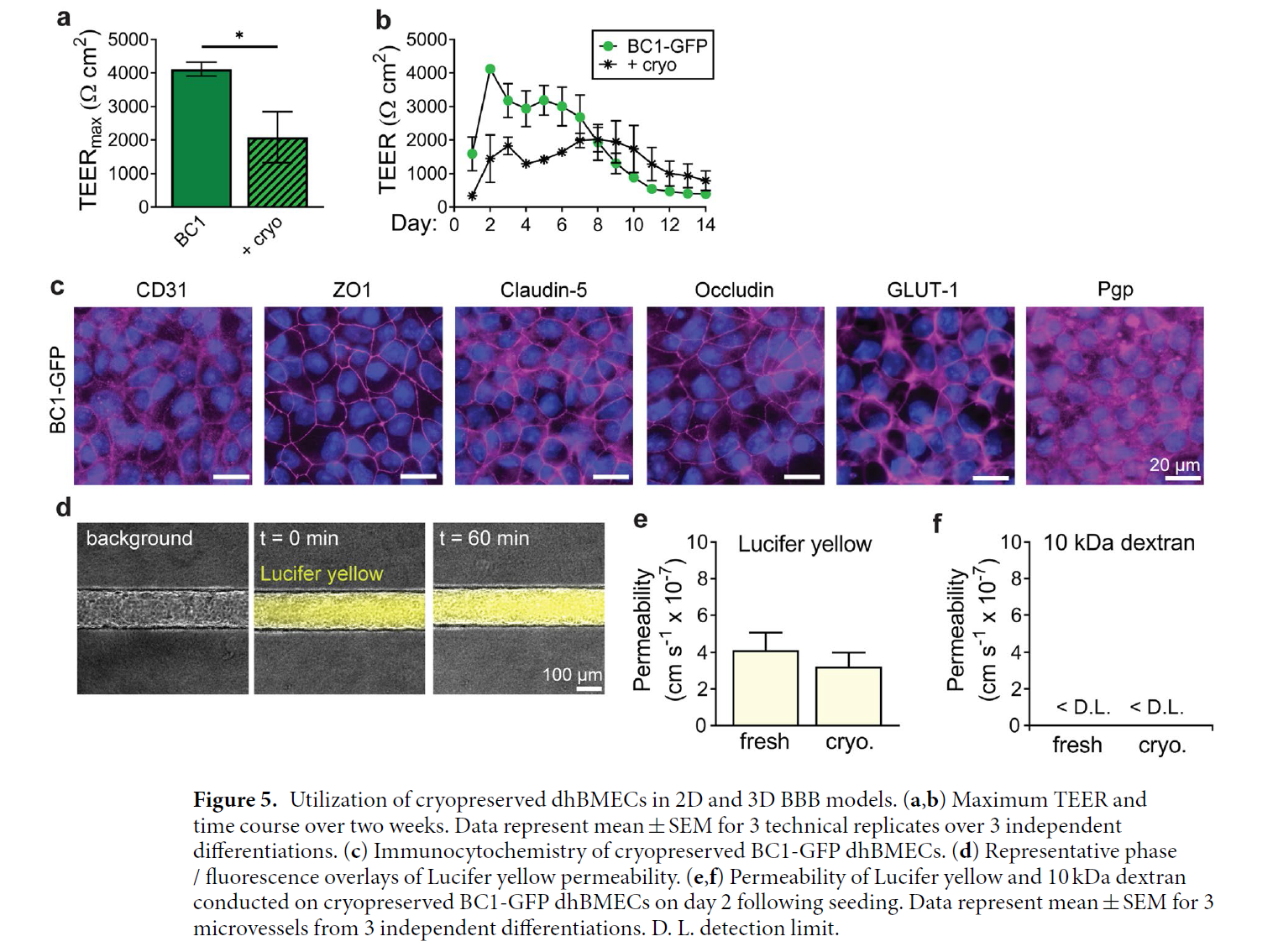
Together, they presented a tissue-engineered microvessels as a versatile platform for studies of BBB function/dysfunction in disease models such as Alzheimer’s disease.
Development Of In Vitro Functional 3D Blood Brain Barrier Model With Tissue Specific ECM Conditions
Thomas DePalma1,2, Hemamylammal Sivakumar 1, Elaheh Rahbar2, and Aleksander Skardal1,2
- Wake Forest Institute for Regenerative Medicine, 2. Wake Forest University, Biomedical Engineering
Brain endothelial cells (BEC) were derived from human induced pluripotent stem cell (hiPSC) according to a previously described procedure [3]. Human astrocytes (AC, ScienCell) were grown on the bottom of a semi-permeable membrane transwell culture system with a pore size of 0.4 um. Human brain vascular pericytes (hVPC, ScienCell) were encapsulated in a hydrogel at a concentration of 5 million cells per mL and seeded on top of the semipermeable membrane. The HyStem hydrogel (ESI-BIO) system was used with UV crosslinking. Staining for tight junction marker ZO-1 and permeability testing using 5 kDa dextran tagged with FITC was used to confirm BBB function. Trans-endothelial electrical resistance (TEER) was measured using an epithelial ohm meter (World Precision Instruments).
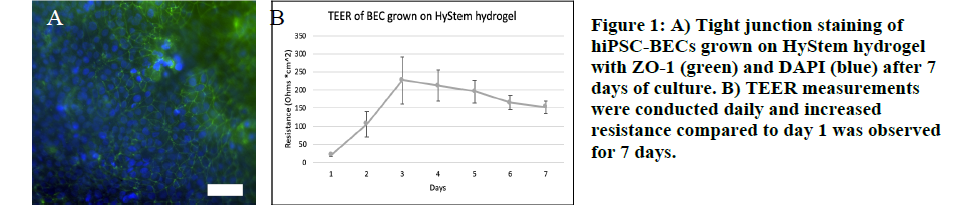
For their future work, they mentioned using hiPSC lines derived from patients with specific neurological disorders in order to study a variety of neurodegenerative diseases including Alzheimer’s disease.
Bioengineered Perfused Human Brain Microvascular Network to Mimic Neurovascular Niche
Max A. Winkelman1 and Guohao Dai1
1 Department of Bioengineering, Northeastern University, 360 Huntington Avenue, Boston, MA, 02115, USA
This group used Aim Biotech chips to test the overarching hypothesis that replicating the physiological features of the neurovascular niche in vitro will promote human NSC self-renewal. Specifically, microvascular networks with luminal flow will enhance NSC self-renewal to a larger degree than those without luminal flow.
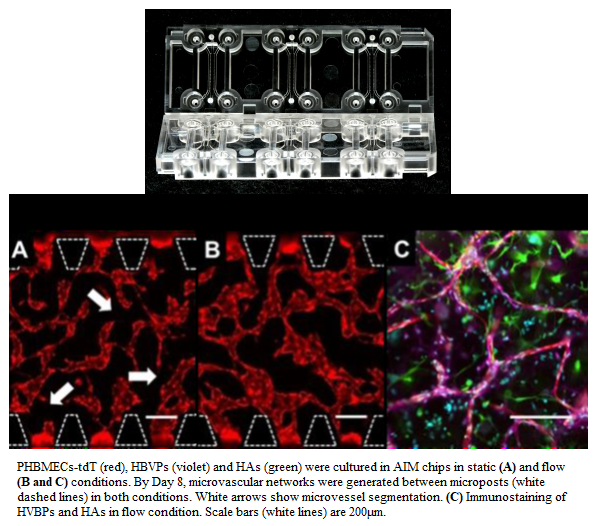
They showed the ability of interstitial flow to generate perfused microvascular networks and reduce microvessel regression within a MFD model. This MFD can also be used to test alternative hypotheses relating to determinants of NSC fate by modifying culture conditions.
An Openable Microfluidic Blood-Brain Barrier: From Artisanal Prototype to Mass Production
Eric C. Spivey1,2, Jacquelyn A. Brown1, Nicole D. Muszynski1, David K. Schaffer1, Shayan Gholizadeh3,
Marcela Mireles3, Thomas Gaborski3, John P. Wikswo1,2
- Vanderbilt Institute for Integrative Biosystems Research and Education (VIIBRE), 2. Department of Biomedical
Engineering, Vanderbilt University, 3. Department of Biomedical Engineering, Rochester Institute of Technology
Our collaborators in Vanderbilt University are trying to develop microfluidic neurovascular unit (NVU) that replicates the BBB, and they presented on the development of an openable, modular NVU (“puck”) that both allows more detailed analytical techniques to be used on cells that form the BBB, and which can be assembled easily with mass-producible parts.
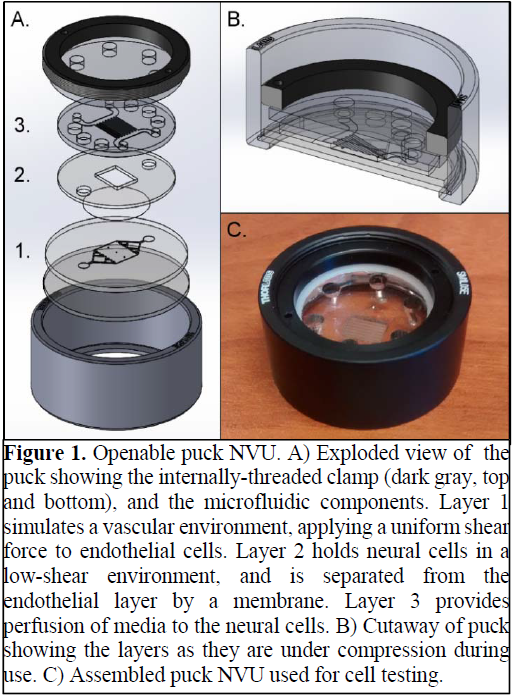
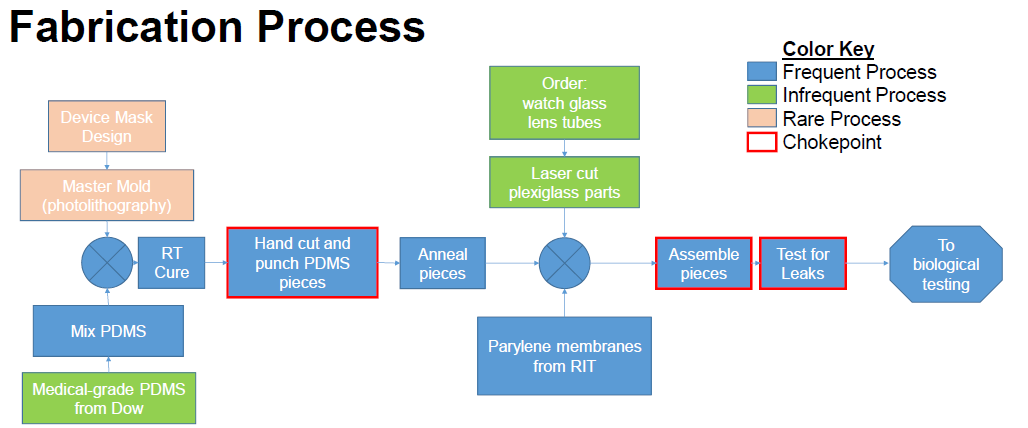
The openable NVU supported the growth of endothelial cells under shear flow on one side of the central membrane, and glial cells on the other side for up to a week. Cells were confluent on both sides of the membrane, and were visually comparable to cells grown in a standard (bonded) NVU at that time point. Ongoing development of membrane materials and preparation protocols is underway to extend the usable lifetime of the puck NVU to >60 days. Tests of puck layers 1&3 made with heat-embossed and/or injection-molded COC show that they are viable mass-producible alternatives to PDMS soft lithography.