Sepsis and Sepsis-Associated Neutrophil Dysfunction – BMES 2019
Introduction
Sepsis related projects had a decent presence at this years Biomedical Engineering Society (2019). I will detail a few of the talks I found most relevant to our group and research.
Nanomedicine for Acute Criticial Illness by Dr. Jacob Brenner (U Penn Medical)
Dr. Brenner is a practicing physician working on ICAM-1, PECAM-1, and neutrophil (moiety proprietary) targeting liposomes (Figure 1) for effective drug targeting to the lung in acute illness like ARDS and sepsis (Figure 2).


The overarching justification for the work is the lack of effective ways to delivery drugs directly to the lung, citing a decrease in drug delivery when lungs are in a state of edema as is the case of sepsis and ARDS. This area of research is particularly important to us, as drug delivery is a logical next step for my work, especially when focusing on luminal versus abluminal delivery of therapeutics. Interestingly, Brenner’s new work (not published) is focused on targeting neutrophil dysfunction in these diseases, something we study in our lab. While some of the specifics were left out of the talk, it appears that Brenner’s lab has developed a small molecule drug aimed at halting neutrophils prior to inappropriate transmigration to the alveolar space. He also mentioned that a lot of the mechanisms behind the effectiveness of the nanocarrier/drug are unknown. Given the opportunity to collaborate, I believe we have a system that would prove extremely beneficial to his work, as well as to the future of our work.
Sepsis Detection – Electrical Profiling of Septic Neutrophils using Microfluidics by Do-Hyun Lee and Integrated Biosensor For Rapid and Point-Of-Care Sepsis Diagnosis by Jouha Min
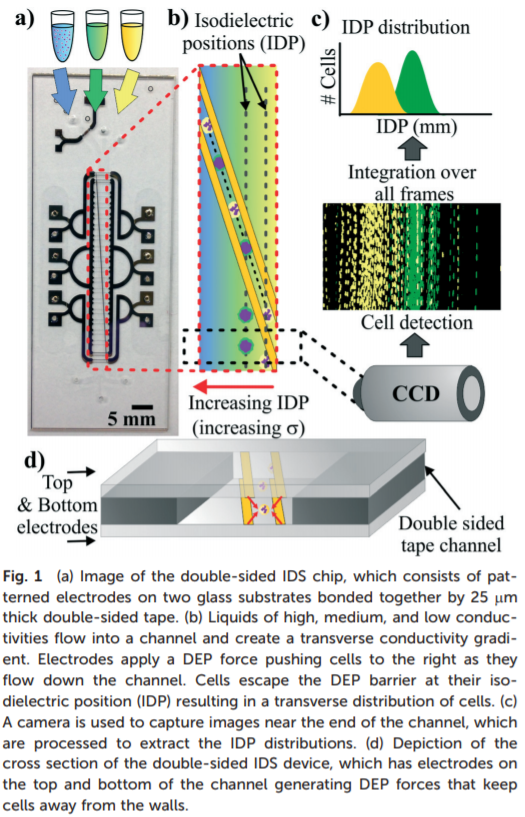
Neutrophils have shown up in many recent papers as biomarkers for sepsis. In “Monitoring sepsis using electrical cell profiling” (Lab on a chip, 2016), Dr. Joel Voldman’s group details a method to monitor sepsis by rapidly detecting neutrophil activation in a microfluidic device. They describe their method as follows: In IDS, a heterogeneous population of cells suspended in high-conductivity media is flowed into a microfluidic chip (Fig. 1a). Other inlets containing intermediate and low conductivity media provide a parallel laminar co-flow. This configuration generates a diffusive conductivity gradient perpendicular to the convective fluid flow (Fig. 1a and b). The channel also contains slanted planar electrodes patterned at the bottom (single-sided IDS) or at the top and bottom (double-sided IDS). An AC electric field applied to the electrodes generates a DEP barrier that guides the cells across the conductivity gradient. Cells escape the DEP barrier at their isodielectric position (IDP), which is the position where they are at dielectric equilibrium with the surrounding medium. Cells with different effective permittivity (or, equivalently, effective conductivity) escape at different IDPs, resulting in a transverse spatial distribution of cells. A camera near the end of the channel takes images of the cells. Finally, imageprocessing software detects these cells and computes the resulting IDP distribution (Fig. 1c).
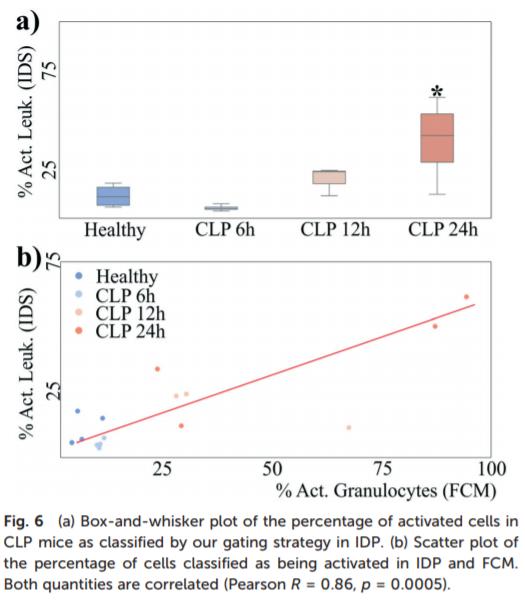
This work is really informative to the activation state of leukocytes during sepsis. Unfortunately, I am skeptical of the specificity of the assay, as no information was given on ICU controls, where you would expect a similar level of leukocyte activation. Additionally, it is clear from their concluding data that this may be primarily a late stage disease marker, and thus not viable in a clinical setting (Figure 3 below) I believe we can still draw a lot from their shortcomings in our own sepsis detection work.
While neutrophils do provide an interesting target for sepsis detection, many groups are still focused on the source of inflammation as a sepsis diagnosis target. In “Interleukin-3 amplifies acute inflammation and is a potential therapeutic target in sepsis” (Science 2015), Dr. Ralph Weissleder’s group identifies IL-3, a regulator of leukocyte production, proliferation, and survival, as an early stage sepsis biomarker. The presenter of this talk, Jouha Min, showed a nice graph highlighting the need for early sepsis detection, as mortality rate of septic patients rises with each hour of delayed diagnosis. In “Integrated Biosensor for Rapid and Point-of-Care Sepsis Diagnosis” (ACS NANO 2018), Jouha Min designed a developed a integrated biosensor device for IL-3 detection from plasma or whole blood (Figure 4 below). The device captures IL-3 on magnetic beads, and a series of processing reactions allow for an electrical current readout and estimation of IL-3 content.

The device showed nice specificity when comparing septic and non-septic ICU patients (Figure 5 below). The exact stage at which sepsis can be detected in this system is a little unclear, but the Science and ACS NANO publications highlight the immense potential of this system, and we should follow this group closely as the sepsis detection work progresses.

Sepsis Associated Neutrophil Dysfunction
Very relevant to our work was the presentation by Brittany Boribong from Caroline Jones’ group out of Virginia Tech. In “Super-Low Dose Lipopolysaccharide Dysregulates Neutrophil Migratory Decision-Making”, Boribong details a microfluidic device with the ability to monitor neutrophil migration towards pro-inflammatory (LTB4) and pro-resolution (fMLP) chemoattractant gradients (Figure 6 below).
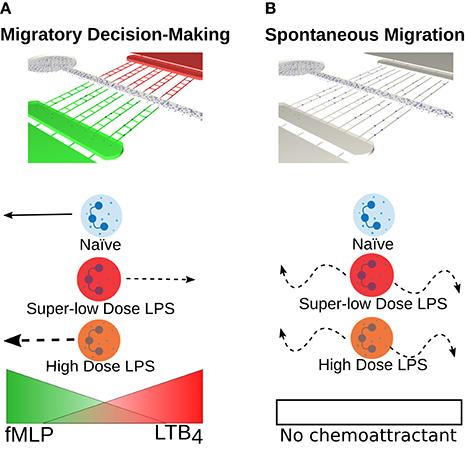
The ultimately concluded that low dose LPS regulates neutrophil migration, analogous to a sepsis phenotype (Figure 7 below). They propose the use of septic neutrophils in future work to repeat this result. While this work does depart from ours, this group does have some nice neutrophil tracking and were very impressed with our phase work on endothelial cells (an obvious next step in their work). We have the potential to repeat this work in our more physiological system. Pre-treating neutrophils and watching them migrate would be fairly trivial and might be a nice direction for our lab. Could be worth reaching out to this group and see if they are interesting in collaborating.