Lung epithelial cells on transwells(1)
A couple weeks ago when I presented at lab meeting, Angela Glading suggested that I look at epithelial cell vacuolization on transwells. This was in order to determine if vacuole formation was endothelial cell-specific. If I’m remembering correctly, the rationale was that the vacuole-less fibroblasts I showed were not contact-inhibited cells and thus not a good comparison for endothelial cells. I had some rat lung epithelial cells (RLE-6TN) frozen from 2 summers ago, so I revived the culture and did some experiments.
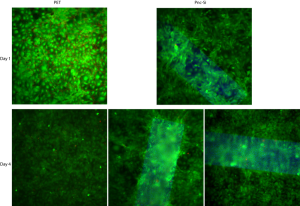
For this experiment, I seeded RLE-6TN at 50000 cells/cm2 on PET and SC612 transwells. I stained with Live/Dead after 1 and 3 days.
After 1 day (top rows), there were a few unstained regions on PET but none on free-standing pnc-Si (there were a couple spots on supported pnc-Si, however). After 3 days (bottom row), there were vacuoles distributed randomly across the PET transwell. It seems like some of those unstained regions are gaps between the cells and not vacuoles, however. On 1 pnc-Si sample, there were no vacoules on or off free-standing pnc-Si, however, a different pnc-Si sample showed excessive vacuolization confined to the free-standing membrane. Based on this first experiment, I would conclude that epithelial cells also produce vacuoles but the process takes longer than for endothelial cells (1-3 days vs. less than 1 day).