Air-blood barrier Images
To follow up my last post on air-blood barrier TEER results, I wanted to show some Live/Dead images of these samples. These are stained after the 14 day experiment.
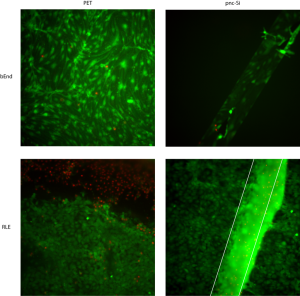
In the following image, you can see normal bEnd morphology on PET and pnc-Si. Only the cells over the pnc-Si slit are visible because these cells were cultured on the well side of pnc-Si. When the RLE cells are cultured on the bottom of PET transwells, there is a pronounced edge effect (dead cell annulus) and a normal, healthy monolayer far from the edges. Importantly, this edge effect seems to be a common shortcoming when cells are cultured upside-down on PET transwells. On pnc-Si, there is blurred fluorescence over the pnc-Si window because there is a multi-layer of RLE cells over the window. I saw some evidence of this at earlier time points (days 1 and 3) in a much earlier post. These cells aren’t randomly clumping like I’ve seen with glial (NG1018) cells, rather they are arranged as a multi-layer. This is exactly the same behavior I’ve seen with fibroblasts. I wasn’t able to get images of the co-cultures for this experiment.
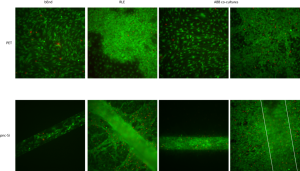
In this experiment, I captured images of co-cultures, as well. Again, bEnd morphology looks normal when these cells were cultured alone (left column). Remember that the bEnd3 cells are on the apical side of the transwells. RLE cells alone on the bottom of PET transwells were interesting again. There was a patchy monolayer on PET, similar to the last experiment. There was a pronounced multi-layer over the pnc-Si slit, but cells seemed to spread from the pnc-Si window in a well-developed quasi-3D network. I’ve seen endothelial cells organize into these types of networks in certain experiments, as well. For co-cultures on PET, I stained the bEnd side (3rd column) and the RLE side (last column) and both cell types looked normal. For co-cultures on pnc-Si, I tried to stain the bEnd3 side (3rd column) but the dye also stained some RLE cells over the slit and the bEnd cells can’t be resolved through this fluorescence. RLE cells on pnc-Si with the co-culture formed a monolayer on supported pnc-Si but a small multi-layer on free-standing pnc-Si. However, the multi-layer doesn’t seem as well-developed (and there is no network formation) as when RLE cells are cultured alone. Could this mean that the bEnd3 cells are actually signaling the RLE cells to brach into a network? I doubt it because: monocultures of bEnd3 cells formed tubes and networks on certain samples when I cultured them long enough, so the same could be true for epithelial cells (since epithelial cells also form tubes in vivo). It’s also possible that RLE cells proliferate on free-standing pnc-Si because of local permeability, so the presence of bEnd3 on 1 side of the slits in co-cultures acts to block this permeability and multi-layer/network formation.
Now, to explain the transient increase in TEER for RLE monocultures and ABB co-cultures shown in my last post. I believe the large increase in TEER is due to the dense, well-developed multi-layers of RLE cells. If you notice in these multi-layers, there are a lot of dead cells after 14 days of culture. It seems like these multi-layers are restricting access to the ‘encapsulated’ cells in these multi-layers. This restriction is causing many of these encapsulated cells to die, which forms paths for diffusion, and thus, decreases the TEER as the cultures age.