Shear-free chemotaxis in action
Hi all, this report will serve both as an update and a template for my upcoming BMES 2012 poster on the shear-free chemotaxis system.
Introduction: Because of its capability in hosting reactions and transporting materials in small volumes, microfluidics enables the efficient processing of precious reagents. However, the shear stress inherent in the use of microfluidics is not desirable at times. For instance, it is difficult not to impose shear stress on delicate cells when fluid flow is used to expedite the exchange of nutrients and wastes and the harvesting of the cellular products of interest. To this end, we have created a microfluidic system integrated with a unique hybrid membrane made of a thin sheet of porous nanocrystalline silicon (pnc-Si) and a silicon nitride (SiN) supporting grid. The diffusion of molecules across the membrane is well less than a second because the membrane is only 15 nm thick, yet the fluid flow across the membrane is significantly attenuated because most of the through pores within the membrane are less than 30 nm in diameter. For this particular presentation, we induced the chemotaxis of cells under a flow-generated chemoattractant gradient, then labeled and washed the migrated cells without detaching them.
Device Fabrication:
The shear-free chemotaxis system consisted of three main components: the flow compartment, which is a polydimethylsiloxane (PDMS) -based gradient generator, the hybrid membrane, and a cell-hosting compartment, all of which are covalently bonded using ultraviolet-ozone treatment (Figure 1). The gradient generator is made with PDMS patterned on a SU-8 mold. The hybrid membrane is fabricated using a combination of standard silicon photolithography and the annealing of a thin layer of amorphous silicon sputtered on a silicon chip. The cell-hosting compartment is made from a custom-cut silicone gasket bonded onto a glass slide.
Figure 1. A. Top view schematics of the components of the shear-free chemotaxis system. From left to right, which corresponds to the order of assembly from top to bottom: the gradient generator, the hybrid membrane, the cell-hosting compartment, and the assembled system. B. Full view of the shear-free chemotaxis system. C. Magnified view featuring the gradient generator and the hybrid membrane.
Cells were seeded in the cell compartment and imaged under a phase contrast microscope at the unset of gradient generation. Two source syringes (0 and 40 nM fMLP) were each pushed at a flow rate of 0.25 uL/min to generate the gradient, which is ~ 0-40nm over 500 um. (A total of 0.5 uL/min for the two syringes combined).
Post-hoc cell-tracking analysis showed that the cells are indeed migrating directionally in the cell compartment and unaffected by the shear-stress created in the flow compartment.
A display of all the tracked cells: [cell_ID] PRESCREEN shearfree_chemotaxis (2012-10-09)
A spiderweb plot of cell migration: [spiderweb_plot] shearfree chemotaxis (2012-10-09)
A histogram on the number of cells migrating in the different directions:
A radial histogram on the statistics of cell migration: [migration radial histogram] shearfree chemotaxis (2012-10-09)
Spiderweb plot and the radial histogram superimposed: [superimposed migration stats] shearfree chemotaxis (2012-10-09)
Shear-free Labeling Experiment:
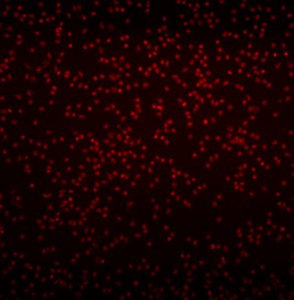
After the cell migration experiment the cells were labeled with a gradient of rhodamine 6G (R6G).
The 40 nM fMLP syringe is switched to a 10 uM R6G syringe and pushed at a flow rate of 10 uL/min (a total of 20 uL/min for the two syringes combined).
Establishment of the R6G gradient: [red] 1. shearfree_labeling_start (7.5 min)
The flow rate of each syringe is then slowed to 1 uL/min (2 uL/min total) and continued for another 10 min.
Continued Labeling with a steady R6G gradient: [red] 2. shearfree_gradient_labeling (10 min)
The flow is then stopped for us to assess how quickly the gradient dissipate/average out.
R6G gradient dissipation: [red] 3. shearfree_labeling_dissipation (6 min)
The 10 uM R6G syringe is then switched to a syringe containing no R6G to clear the R6G from the system (at a total flow rate of 20 uL/min).
R6G clearance: [red] 4. shearfree_labeling_clearance (10 min)
At last, an image of the labeled cells: