Fluorescence on Single-Well CytoVu – Phalloidin & DAPI
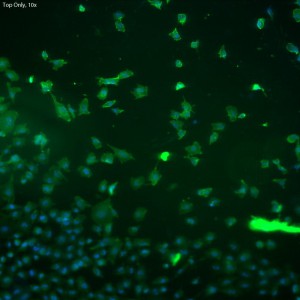
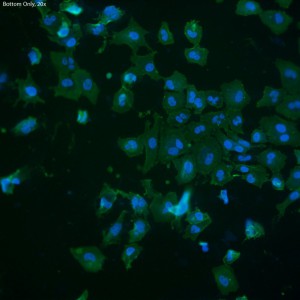
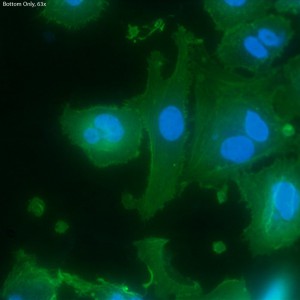
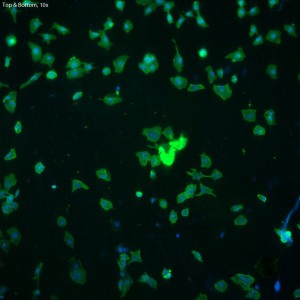
In this trial, bEnd.3 cells were seeded onto three single-well CytoVu devices with SiO2 membranes coated with a 1% Geltrex coating. After ~27 hours, the cells were fixed and permeabilized (the fixing may be only partial though, as the solution used was more than 5 months old). After this, DAPI and FITC-conjugated phalloidin were used to stain the cells’ nuclei and actin, respectively. Stain was applied in one of three methods: only on top of the membrane (the side with the cells), only on the bottom of the membrane, or on both top and bottom. It seems that any of the three methods is effective, as all produce properly stained and bright cells.
The cells all appear viable and predominately spread, showing that the Geltrex coating on oxide membranes does encourage healthy cells. Whether or not the cells are physically interacting with the pores themselves is difficult to determine without higher magnification or use of a different microscopy technique (such as SEM?). Even with solely the fluorescent images, it does seem possible that there is interaction between the pores and the leading actin filaments.
 The focal depth in this image shifts from top to bottom,
The focal depth in this image shifts from top to bottom,
possibly caused by either a warping of the membrane due
to the cells, or by the bolus of fluid on top of the membrane.



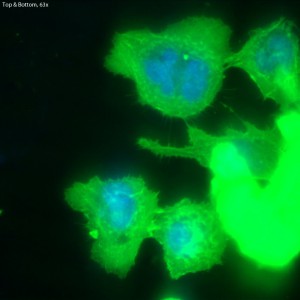


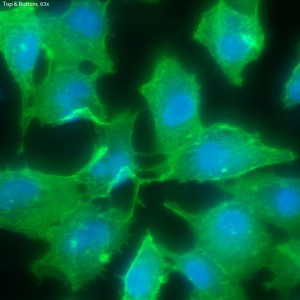
is this done on an inverted or upright microscope?
What was your integration time? On what kind of camera? (i assume this is wide-field rather than confocal?)
The microscope is inverted and wide-field, yes, a Leica DMI6000.
The camera used is a cooled EMCCD, with exposure times of 90ms for the DAPI, and 600ms for the FITC (phalloidin). EM gain was used as well to help boost the signal.