NP separations with NPN using constant pressure setup
Our recent work using NPN to separate free IgG from 20 nm NPs has shown that we can pass liquid under either forward or reverse configurations using the centrifuge. We had thought that there would be greater resistance in the forward orientation due to cake layer formation. We had found high resistance to flow in high concentration solutions using a constant pressure setup in the past, so we assumed that the forward centrifuge would have similarly low flow.
To better understand the different behavior for centrifuge vs constant pressure, we attempted separations of 20 nm NPs ( no IgG) using the constant pressure setup. The plot below shows that flow is dramatically reduced under constant pressure for 10 minutes for higher concentrations. (I’ll need to complete the study using all of the concentrations) Using the constant pressure cell also allows for the qualitative observation that most of the filtrate passes in the first moments (several seconds) after applying pressure.

I tried to measure fluorescence of the filtrates to see if NPs were being retained by the filter and got the following spectra:
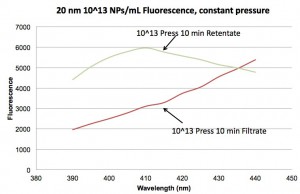
Unfortunately, there seems to be a background fluorescence contaminating the filtrate signal, so I can’t be sure if the NPs are passing the membrane or not. I see the same “contamination” in another filtrate curve, albeit at different intensity. I don’t see this contamination in any of the retentates (solution in SEPCON assembly), or starting solutions (in 2 mL centrifuge tubes), or baseline measurements using only buffer (also in 2 mL centrifuge tubes). So my best guess is that the contamination is coming from an interaction of the filtrate with the 1.5 mL centrifuge tubes. I’ll need to test this independently. I’m hoping we haven’t noticed this before because we have generally worked at higher concentrations of NPs, closer to 10^15, so any contaminant may have been overwhelmed.
Our gold separations have been done in the pressure cell at concentrations of ~10^12 particles per ml and below. So the fact that you are beginning to see the pressure cell flowing for PS at these concentrations seems consistent with the experience with gold.
It would be good to see 10^11 in this plot.
How well are we matching to the pressure in the centrifuge?
Interestingly gold, being much heavier than water, pellets in the centrifuge but not in the pressure cell. So the pressure cell might be the right solution for metal NPs while the centrifuge is the right solution for plastic (and lipid?) NPs.
I did try 10^11 and the entire volume passed after approximately 30 s. The membrane looked intact, so the high flow rate is possibly correct.
According to the sheet on the centrifuge, 3000 rpm is equivalent to 16.7 psi. If I try to apply 15 psi, the membranes tend to break immediately. So I backed off pressure to 5 psi which generally did not break the membranes. I have started a couple membranes at 5 psi and ramped the pressure up to 10 and 15 psi to see if I could induce flow. As I increase the pressure, I do not see an obvious change/increase in flow. It may be possible to reproduce this data by starting at a low pressure and quickly ramping up, but I think a lot of the total flow will occur during the same time that we are ramping up.