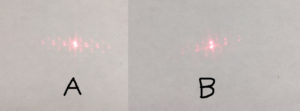Bead Aggregation Experiments (Beads + Protein)
Here are the first experiments I have performed with the SepCon slots, under the guidance of Jamie and Kilean. The goal of these experiments is to prove that goat anti-human beads (6.8 um diameter) can be combined with a low concentration of Human IgG (1 mg/mL) which will cause aggregation, and therefore accumulate on the surface of a membrane with 8-9 um slots.
We setup two controls: the first is passing beads through with no protein present. This was successful, we only had a handful of single outlier beads trapped in the slots out of the approx. 66k that were passed through. The second control was to combine goat IgG with the beads, however there was enough similarity between goat and human IgG, that we still formed aggregates, therefore, we will try BSA instead (has not been done as of this post).
Experimental conditions:
First the beads were diluted, 50 uL stock solution in 950 uL PBS
Human and Goat IgG were prepared at 1 mg/mL in PBS and the remainder was stored in the freezer.
All experiments were allowed to mix for 20 minutes at RT. The samples were then loaded into SepCons and either gently tapped or spun at 500 RPM for 2-5 minutes (the low RPM was not needed, but allows for more consistency, so this will be the method going forward).
1) 1 mL PBS, 50 uL dilute bead solution
Result: As expected, only beads left on surface were too large to pass (larger than 9 um).
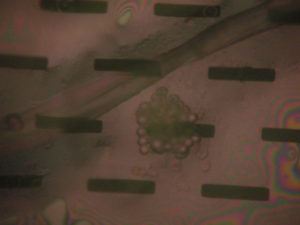
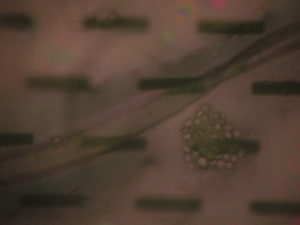
2) 990 uL PBS, 50 uL dilute bead solution, 10 uL Human IgG
Result: Aggregation was achieved, however it was sparse, < 1% of slots were filled. See Images
3) 990 uL PBS, 50 uL dilute bead solution, 10 uL Goat IgG
Result: Unexpected, Goat IgG bound to the beads mimicking the Human IgG
4) 980 uL PBS, 50 uL STOCK bead solution, 20 uL Human IgG
Result: Too. Many. Beads. Could not distinguish between aggregates and the general cake on the slots and on the 0.5um secondary filtration (see below).
5) 980 uL PBS, 50 uL dilute bead solution , 20 uL Human IgG
Result: Significant improvement over 10 uL, many more aggregation sites, however still relatively few ‘clogged’ slots.
6) 960 uL PBS, 50 uL dilute bead solution , 40 uL Human IgG
Result: Almost filled all slots, still some open so we might have to tweak the bead count and/or protein levels.
For experiments 5&6, I tried to capture the beads from the ‘waste’ but both membranes broke and I saw very few beads. I will repeat these and try to determine how many beads we are ‘consuming’ to get an idea how our ratio of protein to bead ratio is functioning. According to Jamie’s numbers, we have about 4400 Protein molecules : 1 bead (66k beads in the dilute 50 uL).
I then tried shining a laser through a fresh 8um slot SepCon and through the 40 uL Chip (#6) and the result was clearly different, see image below, A is the fresh chip, and B is the chip with aggregates on it…sorry about the crude identification (insert rant about using a Mac).
Kilean has also tried a 7th experiment:
50uL dilute bead solution, 500 uL whole blood (Heparinized)
Result: many aggregation sites. See Images