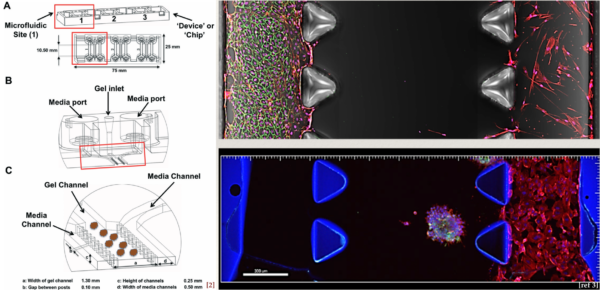
BdEC/T24 co-culture: invasion assay (preliminary)
Introduction
Motivation: We want to assess if the bladder cancer cell line T24 would influence the baseline characteristics of bladder epithelial cells (BdEC, a.k.a. ATCC® PCS-420-010™) when the two cell types are cultured together. Ultimately, we are interested if the T24 induces BdEC malignancy (turning the normal epithelial cells cancerous).
Goal: Investigate if BdEC monolayer will break away and invade into ECM gel toward and T24 cells.
Factoids: I first tried growing the BdEC cells in membrane inserts that are either coated with geltrex or bovine collagen I (after 2 hr incubation in the incubator). For geltrex, I used the stock straight up at 150-200 μL/cm^2. For the bovine collagen I, I used 2 mg/mL at 150-200 μL/cm^2. However, the BdEC are not able to spread to form a monolayer on the coated membrane even after a week. I sort of knew this may happen since the BdEC is not very adherent in general.
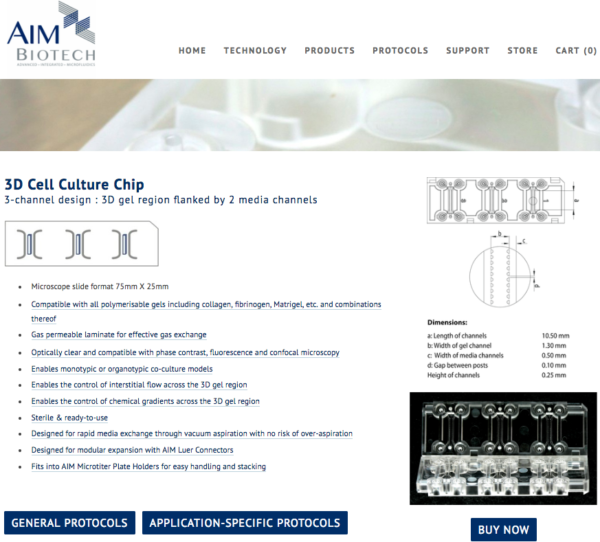
One solution around this is to have the invasion occur laterally, while culturing the BdEC on hard substrates. Out of interest of time, I decided to buy instead of making one in house. I also wanted to get a feel of the commercial devices, assessing the pros and cons. I decided to go with devices sold by AIM Biotech (Roger Kamm is affiliated with this Singapore-based company):
These devices are priced at a pack of 25 chips for $375. Each chip contained 3 devices ($5 per device).
In terms of use this device is not shabby. The user manual is very well-written. However, the device could use some QC since the bonding between parts does not seem all that great.
I used p10 pipette to introduce the Matrigel into the middle channel. Of the three chips (9 devices total, two had leakage of the Matrigel into the surrounding channel). This could be me… but I am usually pretty careful with pipetting in general.
These are some observations:
1. On gel introduction. I introduced 2o μL of the Matrigel stock (concentration of 11.9 mg/mL) into the middle channel and waited for 2 hr in the incubator for gelation (most protocol recommend at least 30 min incubation for Matrigel). The manual also recommended that for new user, it is better to split the gel introduction into two repeating steps, 5 μL each time from a different port. The double introduction from different ports is intended to reduce gel spillage into the flanking μ-channel. I did the gel introduction in one shot of 10 μL, and observed spillage in 2 out pf 9 devices. I will resort to the two step gel introduction next time.
UPDATE: In attempt to reduce spillage of the Matrigel from the middle channel into the flanking μ-channel. The spillage is likely due to high viscosity of the Matrigel. We knew this may be the case since the pipetting of the Matrigel feels more “viscoelastic”. Furter more, 2mg/mL of collagen I always work (versus the 11.9 mg/mL of the Matrigel stock). Stephanie and I tried 1:1 dilution of the Matrigel with PBS and that currently has near 100% success rate.
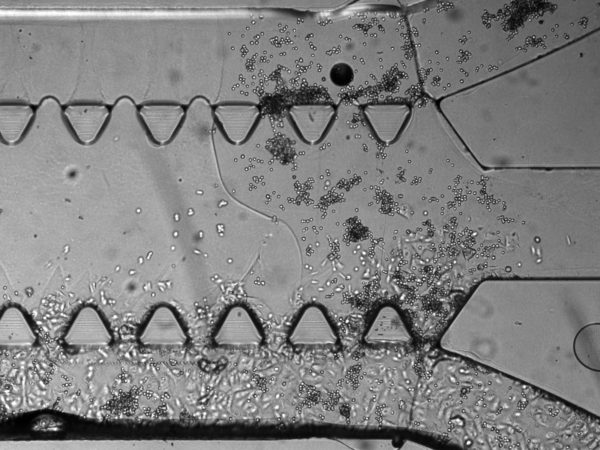
2. On cell seeding. The manual recommend seeding the cells at 1.5 x 10^6 cells/mL. This is not new to us since in general we need to up the cell concentration (due to the large surface area to volume ratio of μ-channels). The manual also recommended seeding at 10 μL, twice, each time from a different port of the same channel. The double-seeding from different ports is likely due to some portion of the fluid moving through the normally gel while flowing tangentially along the μ-channel. We do indeed see lower cell density in the middle of the channel: 
2. On poor QC. I also saw some device defects, notably the presence of “bubble” suggestive on bad bonding between parts. I happened to see one case in which the bad bonding led to leakage of cells outside the flanking μ-channel.
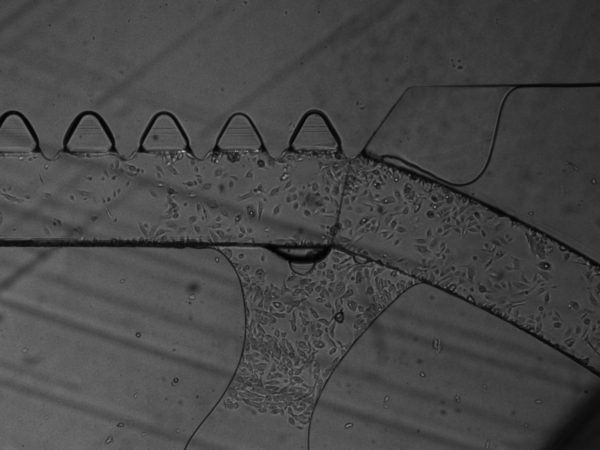
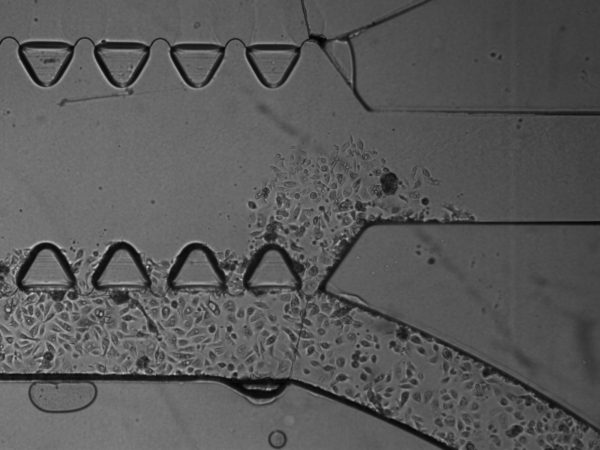
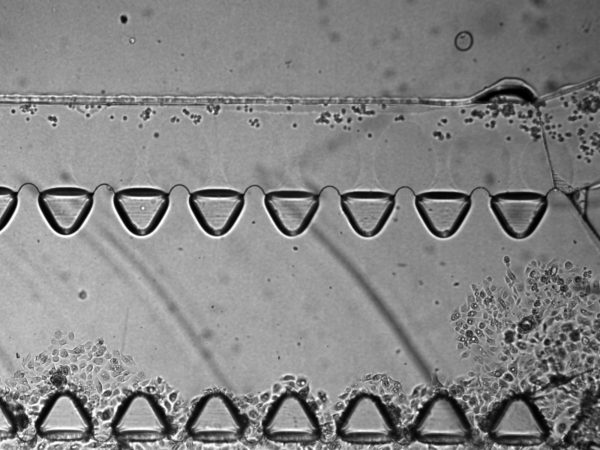
3. On BdEC invasion. Over time, I also saw overgrowth of the BdEC though the gel, even though I have not cultured cells on the opposite μ-channel:
Coincidentally (or maybe not), this always happen near the entrance side where the fluid flow through the gel is the strongest. Since serum is a chemoattractant to BdEC. I will use PBS on the opposing μ-channel to reduce potential BdEC chemotaxis.
4. On BdEC adhesion/spreading. I also coated the μ-channel with 1:100 dilution of Matrigel. I saw BdEC spreading as early as 4 hr. This is indicative that BdEC like the Matrigel coating better than the T25/75 flask as usually at least 12 hr is required for notable degree of adhesion and spreading.
5. On evaporation. The manual recommend media renewal everyday. On my past experience it is possible to get away with as little as 20 μL of media volume over the weekend as long as enough moisture is provided. In this case, I place the device in a 10 mm petridish, with two sheets of Kimwipes (rolled into a “pillow”), with each pillow dosed with 1 mL of DiH2O, and rehydration as need (200 μL) every other day. In retrospect, the best way is probably to weight the device each time to monitor for moisture loss and rehydrate accordingly. I used a media volume of 120 μL per flanking compartment (50 μL in each well, with 20 μL in the μ-channel itself). The BdEC did well and was able to proliferate quite well over the weekend (with no media renewal until monday).
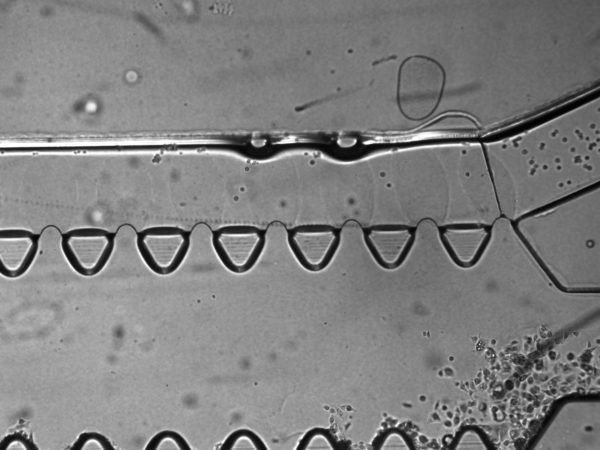
6. On Matrigel fingers. I saw protruding, finger-like structures emanating out from between the triangular pillars, and wondered if these are Matrigel that oozed out over time. It seems that is indeed the case, since the T24 cells that I introduced cannot pass through these fingers (this also suggest that these fingers are also sufficiently tall). In one instance, the Matrigel fingers occlusion of the top μ-channel is severe enough that I even busted through the middle gel as I introduce the T24 cells into the top μ-channel. It is possible that this oozing out of Matrigel fingers is due to MMP degradation of the Matrigel by the BdEC. For my next experiments, I will try acellular control, with media and PBS only, to assess whether this oozing out is due to cells. I will also precoat the middle channel with 1:100 dilution of Matrigel prior to introducing the Matrigel stock to increase the adhesion of the Matrigel to the middle channel.
UPDATE: Stephanie and I ran another set of experiment. For some of the samples there are no cells, and either PBS or the culture medium (PECBM) in the flanking μ-channels. Some of the samples have small fingers but these fingers never grew even after 18 day. We are currently observing samples that have cells seeded into both the flanking μ-channels.
7. Potential Concern: Cells migrating between the gel and the substrate/wall instead of invasion through ECM digestion. For all images the cells invading through the gel are in focus at the substate floor. This raises concern that the cells are migrating underneath the gel rather than digesting though the gel via protease such as the MMP. It is possible that this may be a combination of both physical squeezing or chemical digestion. Furthermore, we always see the cells always break through at the junking near the entrance and the exit of the flanking μ-channels (as shown in many of the prior images). This may suggest that these are the mechanical weak point of the gel adherence to the wall.
I have tried to coat the surface first with 1:100 dilution of the Matrigel, let incubate for 1 hr, then remove and dry the samples in a vacuum chamber for 1 hr. I then introducing the 100% Matrigel stock for gelation. The rationale behind this approach is to let the gel bond to the coated protein. However, the Matrigel that were later introduced has a high frequency of leaking into the μ-channel (even with the 1:1 Matrigel:PBS mix).
8. Device Assembly: Stephanie and I decided to break some of the used chip down to better understanding its making. It seems there are two major parts to the device: 1. A gas permeable substrate (100 μm polymer, maybe COC) and an injection molded top part, which contained all other features of the devices (e.g. the triangular posts and the wells for holding the media). These two parts are taoped together (see image below).
The two parts are also bonded, preferentially at the triangular posts and doesn’t seem to be everywhere. This may explain some of the defects that we sees.
Work in Progress: Automated quantification of BdEC invasion.
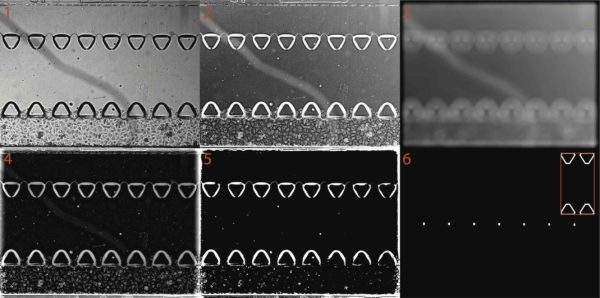
Example of the image processing workflow performed to identify the triangular posts were shown in the figure below:
1: original image
2: flipped image (the darker pixels were converted to whiter pixels, this is done by subtracting the whole image from the image intensity maximum).
3. background image (obtained by blurring the whole image with a disk filter with a radius of 50 pixels). One can appreciate that due to the lack of the uniform illumination, the left side of the image is darker than the right side.
4. background-corrected image (obtained by subtracting the background image from the flipped image. Note that the backgrounf illumination is now much more uniform.
5. binarized image, obtained by assigning all pixels with intensity greater than the 90th percentile value to white (1), while all other pixels were assigned as black (0).
6. template-matched image, obtained by normalized cross correlation. Essentially, a convolution between the binarized image and a template of the triangular posts (shown in the inset).
Example Outputs:
The triangular pillars auto-IDed were makred in blue. The invading cell mass auto-IDed were marked in orange. The 4 images (40X total magnification, image width ~3.376 mm) below completely covers the entire cell culture area of the device.
Full cell culture μ-channel (26 triangular posts, spanning 10.5 mm)