Exploring Formlabs SLA 3D printing for cell culture applications
The initial purpose of exploring formlabs SLA 3D printer for a collaboration with Dr. Nicola Marchi (link to previous post). The purpose of the this work was gaining the ability to use a simple membrane on 3D printed platform for BBB modeling and WBC migration though endothelial (and glial cell layers) and collecting and analyzing them through sampling ports.
Overview of Stereolithography (SLA)
Stereolithography belongs to a family of additive manufacturing technologies known as vat photopolymerization, commonly known as resin 3D printing. These machines are all built around the same principle, using a light source—a laser or projector—to cure liquid resin into hardened plastic. The main physical differentiation lies in the arrangement of the core components, such as the light source, the build platform, and the resin tank.
SLA 3D printers use light-reactive thermoset materials called “resin.” When SLA resins are exposed to certain wavelengths of light, short molecular chains join together, polymerizing monomers and oligomers into solidified rigid or flexible geometries.

Figure 1. Form 3B SLA printer
SLA parts have the highest resolution and accuracy, the sharpest details, and the smoothest surface finishes of all 3D printing technologies, but the main benefit of the stereolithography lies in its versatility.
Material manufacturers have created innovative SLA resin formulations with a wide range of optical, mechanical, and thermal properties to match those of standard, engineering, and industrial thermoplastics. There is wide library available for commercial SLA printers such as Formlabs series printers. Among commercial SLA printers, Form 3B is the first specifically optimized for biocompatible materials.
Initial experiments (Issues with cell attachment and biocompatibility)

Figure 2. Dr. Marchi’s setup for a BBB continuous flow model
Figure 2 demonstrates Nicola’s setup. So the first step to our contribution was to either use the 3D printed parts he sent to us or we made using his design, attaching the membranes on top using biocompatible glues to double sided tape, and shipping (back) to France for him to use.
However, he had difficulties attaching and growing cells time after time. We used the same setup to grow Human Umbilical Vein Endothelial Cells (HUVECs)/Bovine Aorta Endothelial Cells (BAECs) to identify the problem. We got the lukewarm results after a few tries (representative of which are shown in Figure 3).
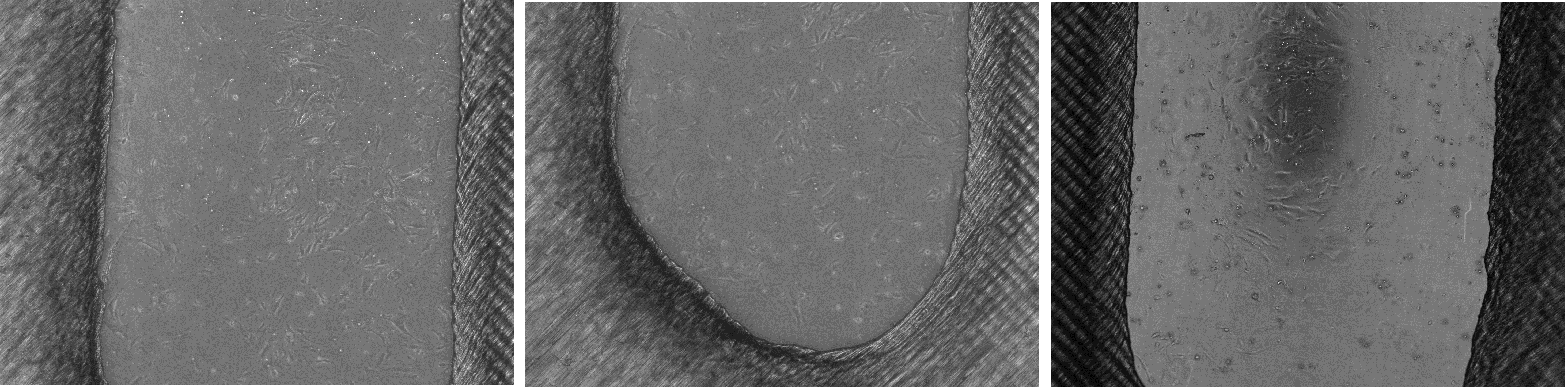
Figure 3. Early representative images of substandard cell attachment on 3D printed devices
Biocompatibility experiments
So we deigned experiments with different conditions to identify what the problem was. The parameters we checked was different washing times (1 hr and 2 hr) and different resins (dental and clear), all left at least 96 hr in room temperature as opposed to previous minimum of 24 hr. The problem was that due to the thinness and fragility of 3D printed parts, we didn’t use standard curing at 70 °C which turned out to be t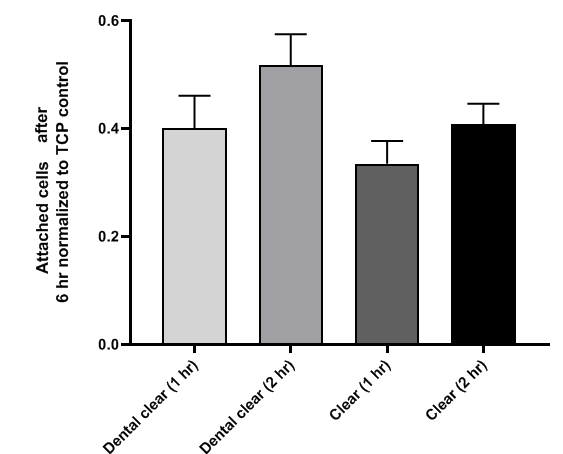 he essence of the problem.
he essence of the problem.
Figure 4. Attached HUVECs after 6 hr of seeding normalized to number of cells on TCP control
Right before the start of the pandemic I tried a different approach and used a modified version of the design for printing with thicker body that can handle the curing. I got really good results with BAECs and the results ended up as part of the preliminary data we presented in my paper.
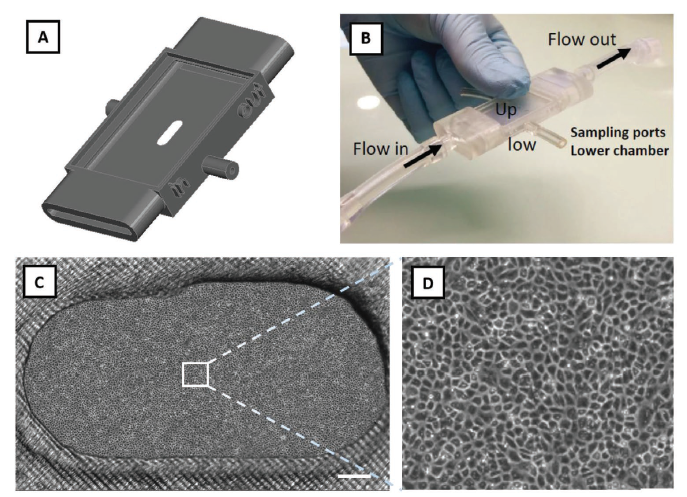
Figure 5. Part of figure 5 of my paper showing remarkable capability of the membrane embedded 3D printed part for growing a confluent layer of ECs using a relatively low density of seeding.
Conclusions
3D printing and SLA in particular offers a variety of opportunities for cell culture applications. The Form 3B printer we have in our lab provides a resolution of down to 25 um which is quite remarkable while also being specially designed for biocompatibility-needed applications. However, these distinctions should be carefully assessed in terms of cell biocompatibility as opposed to intended dental application. There is now a growing number of publications (as opposed to when I started this) specifically targeting the potentials and pitfalls of 3D printing for cell culture devices, which should be thoroughly read and used before conducting cell culture experiments with SLA 3d printed devices.
Interesting paper
Kreß, Sebastian, Roland Schaller-Ammann, Jürgen Feiel, Joachim Priedl, Cornelia Kasper, and Dominik Egger. “3D Printing of Cell Culture Devices: Assessment and Prevention of the Cytotoxicity of Photopolymers for Stereolithography.” Materials 13, no. 13 (2020): 3011.



From Kevin Webb (Nottingham) …
The is interesting stuff. The cytotoxicity data parallels the work we did in Nottingham with Greg’s clamshell design for the nanomembranes (2 slides attached).
At the time we speculated that some incomplete curing or incomplete washout of a photocuring agent might be culpable.
Regarding attachment have you contemplated an FSL overlay?
https://trace-bmps.org/3d-printed-cell-killers/