Little Back Dots in Coated Controls … How We Wonder What You Are.
Introduction
We are investigating some issues seen with long term attachment (up to 6 days) of the EECM-BMEC-like cells seen by collaborators in Bern, where they noticed black aggregates on the coated control devices that get worse at day 6. Some attachment issues and aggregates have also been seen at the University of Rochester in my experiments. These attachment issues could have a number of causes including an issue with the membranes, an issue with the coating solution, or an issue with user technique. This blog post will examine the issue from the perspective of investigating if the issue of the black aggregates is from the membranes themselves.
Results
Experiment 1 – Black Dots Noticed on Coated Control Device
In the first experiment, six devices were seeded with cells and one device was coated but not seeded to keep as a control. The devices had issues growing to confluency. As seen in Figure 1, by day five there were still gaps in the cell layer. This may be due to pipetting too strongly through the channel or introducing bubbles during seeding by a new µSiM user, but since collaborators in Bern were also experiencing issues, this was further investigated.
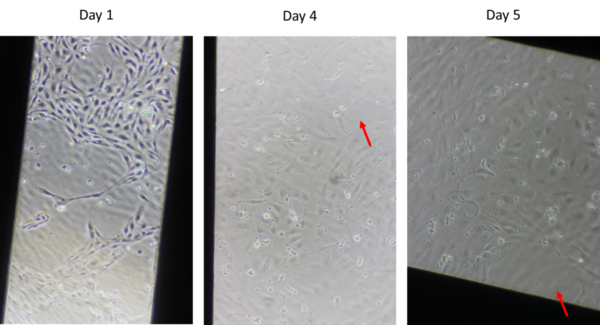
Collaborators in Bern noted black aggregates in the coated control with their devices that had cell attachment issues over time, so the coated control device for this experiment was examined for aggregates. As can be seen in Figure 2, the black aggregates were also present in this experiment and persisted in the coated control over six days, though it does seem that some of the smaller aggregates washed off, leaving primarily larger aggregates. Or perhaps the small aggregates aggregated further into large aggregates.

Experiment 2 – Investigating Lot-to-Lot Variability of the Black Dots
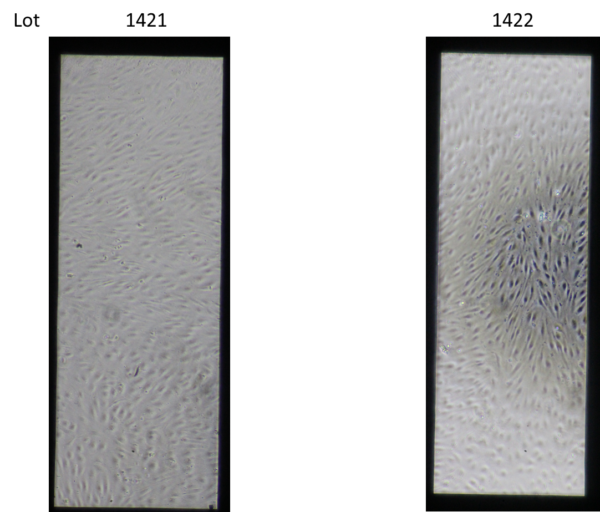
One possible explanation of the black dots is that there were introduced in the process of manufacturing the membranes. This could potentially be due to membranes that we know are not fully etched, insufficiently removing the other materials involved in the manufacturing process. To examine this hypothesis, we looked at membranes from three different lots (1368, 1421, and 1422) because if the issue was stemming from the membranes, we would expect to see variability between lots. One membrane from lot 1368 was included because that is an older lot known to be fully etched, whereas the membranes from lots 1421 and 1422 are not fully etched. When the membranes were examined before they were coated, all of the membranes appeared clean as seen in Figure 3 (we did not take a picture of 1368, but it also looked clean).

The devices were then coated with a solution of 4:1:5 parts human placenta–derived collagen IV (Millipore Sigma, Part # C5533-5MG ): bovine plasma–derived fibronectin (Sigma, Part # F1141): water. After the coated solution was allowed to stay overnight, they were imaged again, and the black aggregates were seen. The amount of black dots varied within each lot and there were devices with both “mild” and “heavy” aggregates in each lot (except for 1368 which only had one device), as seen in Figure 4. These observations suggest two conclusions: one that it is not the membranes themselves causing the black aggregates, but either the coating solution or an interaction between the membranes and the coating solution and two that the cause of the black aggregates is not from a manufacturing difference in a specific lot since there were both mild and heavy aggregates seen in multiple lots.

One clean device from lots 1421 and 1422 were seeded with cells and the remaining devices were monitored to look at their surface over time. By day five, there were overall not significant changes, but the device with “mild” aggregates from lot 1422 had heavier aggregates on day 5, as can be seen in Figure 5.

The cells, which were seeded on “mild” aggregate devices from lots 1421 and 1422 grew to confluence and continued to look healthy until day five, as can be seen in Figure 6.
Experiment 3 – Using Plasma to Treat the Membranes
The next experiment investigated the possibility of using a corona wand to plasma clean the membranes. It was hypothesized that plasma could clean the surface and/or increase the hydrophilicity which could then alter the interactions between the membrane and the coating solutions and potentially limit the formation of the aggregates. To test this the membranes were plasma cleaned before assembly and then coated a few days later. Three membranes from two lots (1421 and 1422) were used and aggregates were still seen on all of the devices as can be seen in Figure 7. The aggregates varied from mild to heavy even within the same lot, as seen in experiment 2.

Experiment 4 – Plasma Cleaning and Different Coating Solutions
Since the plasma cleaning did remove the black aggregates, two more questions were investigated in the next experiment. The first was whether waiting between cleaning the devices and coating them in Experiment 3 led the plasma treatment lose its effect and the second was whether using a different coating solution would prevent the aggregates. To test the first question, in this experiment the membranes were treated with plasma, assembled into devices and coated on the same day. To test the second question, an alternative solution was used to coat the devices made of 100µg/mL rat-tail collagen type 1 (Sigma, Part #: C3867-1VL) and 18.5µg/mL of human fibronectin (R&D Systems, Part #: 1918-FNas) compared to the original solution of human collagen type 4 and bovine fibronectin. As can be seen in Figure 8, plasma treating, assembling the devices, and coating all in the same day did not prevent the formation of aggregates with the original coating solution (hCol 4/bovine Fn). On the other hand, the alternative coating solution (Rat Col 1/ hFn) seems cleaner that the original coating solution. As can be seen in Figure 9 which zooms in on the third Rat Col1/ hFn image from Figure 8, there is the faint formation of some sort of fiber in the alternate coating solution, but it is not as significant as what we see in the original coating solution.
There are a couple of factors which complicate this conclusion. The first is that the human collagen type 4 was expired by the time this experiment was performed and the bovine fibronectin was almost two months old. However, this was not an issue when the first experiments were performed, and the aggregates formed on those experiments as well. Also, the rat collagen 1/human fibronectin solution only needed 1 hour to coat whereas the human collagen 4/bovine fibronectin solution needs overnight coating, so the differences seen might be from the length of the coating and not the solutions themselves.

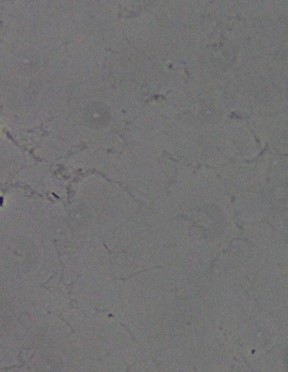
To see if this issue was unique to the membranes used in conjunction with the coating solution or if the aggregates were just from the coating solution itself, some wells of a 96-well plate were also coated. As can be seen in Figure 10, no aggregates are seen, but it is difficult to determine if this is because there were no aggregates or because the imaging is not good enough on the 96-well plate to pick them up.

Figure 10: No aggregates can be seen on a 96-well plate.
Conclusions and Next Steps:
Overall, these experiments demonstrated a couple of points. One is that the black aggregates can be seen here at the U of R as well as in Bern. Another is that they are not on the membrane surface until after the coating solution is applied, suggesting that they are from the coating solution or from the interaction between the coating solution and the membranes. Since the membranes should not interact with the coating solution, because SiN is not very reactive, it was hypothesized that this was due to a change in the manufacturing process leading to the membranes no longer being fully etched, which could mean there are other molecules left on the membranes which can interact with the coating solution. This experiment tested this by comparing a membrane from before the change in the manufacturing process and after, and the black aggregates were seen on both, leading to the conclusion that the change in the manufacturing process is not the issue. It was also seen that there is variability within each lot regarding how many black aggregates form, indicating that the issue is not merely one bad lot. Cells did grow to confluence and stay healthy until day five on membranes with a mild amount of black aggregates, showing that at least in limited quantities the aggregates do not interfere with cell health.
Based on these results, we investigated treated the membrane surface with oxygen plasma to clean the surfaces and/or increase the hydrophilicity can alter the interactions between the membrane and the coating solution and limit the formation of these aggregates and lead to better cell health. Unfortunately, the plasma treatment seemed to have no impact on the formation of the black aggregates. We also looked into an alternative coating solution which did not form the black aggregates, although it did form some faint fibers, which indicates that the coating solution is the source of the fibers. Our collaborators in Bern also saw the black aggregates in the coating solution and that same coating solution made by preparing the collagen in water instead of acetic acid. However, they were able to culture cells to confluency by day 6, using a different membrane lot than they had previously. This shows that cells can be successfully grown on devices with mild black aggregates, so there is some variability in this data, and the actual cause of the difficulties growing the cells to confluence is inconclusive.
The next steps in this experiment are to further look into ways to limit the formation of these black aggregates on the membranes. One thing to look into is if there is a difference in the aggregates seen with new reagents compared to older reagents. Another is to look further into alternate methods to clean the membranes. While the plasma treatment did not work, cleaning with a chemical solution such as ethanol or piranha cleaning the membranes may be more effective. It is also important to focus on solutions that allow for the cells to grow to confluence and not focus on the black aggregates specifically, since we have seen that the cells can grow to confluence with some aggregate formation.
Follow Up 1:
Experiment 5 – Ethanol Cleaning Membranes
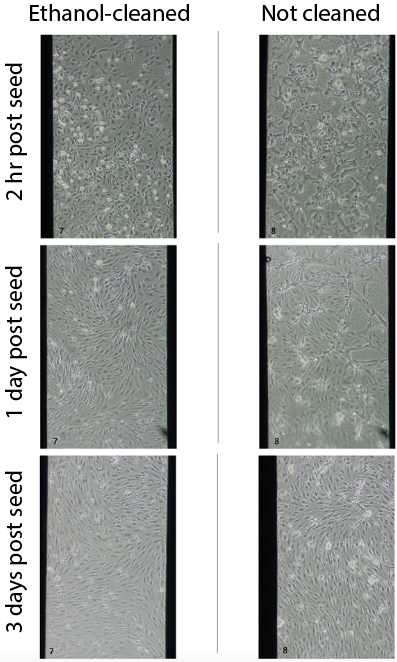
Since the plasma cleaning did remove the black aggregates, we tried ethanol cleaning to wash off debris. After assembling and UV-sterilizing devices, just prior to coating, we added 50 µl 70% 200-proof ethanol to the well for a few minutes, washed with 100 µl sterile, UltraPure water, and then added the coating solution (EECM-BMEC solution, hCol4/bFn). For this set, I ethanol cleaned 3 devices and did not wash at all the remaining 6. All ethanol cleaned devices had minimal aggregates formed, the uncleaned devices had a lot of aggregate formation.

All 3 ethanol-cleaned membranes had good cell attachment, growth, and visual health. Only 2/6 uncleaned membranes had comparable-looking cells. The rest had cell attachment and growth but the monolayers did not look as healthy, as shown in image below.
I ran permeability on 1 ethanol-cleaned and 2 uncleaned membranes. Ethanol cleaning appeared to improve permeability of EECM-BMEC-like cells.
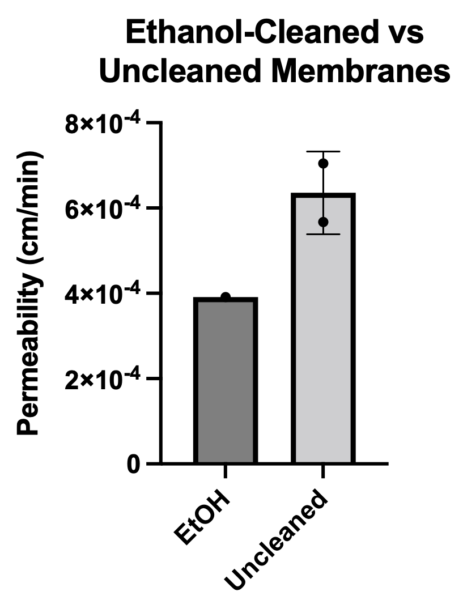
![]() Experiment 6 – Ethanol vs. Water Cleaning Membranes
Experiment 6 – Ethanol vs. Water Cleaning Membranes
To confirm that ethanol is necessary for cleaning the membranes, rather than just a wash step, I did a side by side ethanol vs water clean. The process was the same as above except for the water clean I used 50 µl water instead of ethanol. For this set, I ethanol cleaned 4 devices and water cleaned only 2. All ethanol cleaned devices had minimal aggregates formed, and the water cleaned devices had some but not a lot of aggregate formation.

Cell attachment, growth, and visual health was comparable between ethanol and water cleaned devices.

I ran permeability on 1 ethanol-cleaned and 1 water-cleaned membrane. Both cleaning techniques produced tight barriers on EECM-BMEC-like cells.
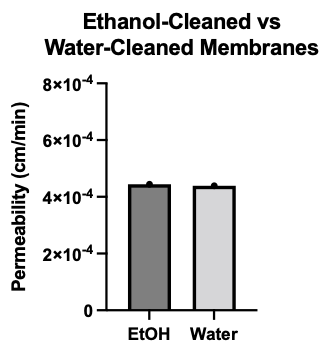
New Conclusions:
We recommend ethanol or water cleaning membranes prior to coating cells. While we do not visibly see the same issue with other coating solutions, it is likely there is still debris on the membrane that may affect the cells. I only washed the side that I added cells to (i.e. top well), however, it may be necessary to wash both sides of the membrane for co-culture, or the channel for channel cell culture.


Data acquired since this experiment was a little less clear. We still get some aggregates even with washing and can achieve permeability <0.5 x 10^-3 cm/min on membranes with a lot of aggregate formation. Regardless, we recommend washing still.
Follow Up 2:
Experiment 7 – Ethanol Cleaning Prior to Assembly
To see if time in the gel box was potentially part of the debris issue, we ethanol cleaned chips prior to assembly, washed with water, and stored in gel boxes for 3 weeks. Then we assembled and coated the devices. We did a side-by-side comparison to chips that were washed post-assembly, just prior to coating. Ethanol cleaning prior to assembly was not effective. This could either come from time in the gel box or from the assembly process. The ones cleaned after assembly, pre-coating still have some aggregate formation but less than the pre-assembly cleaned chips.
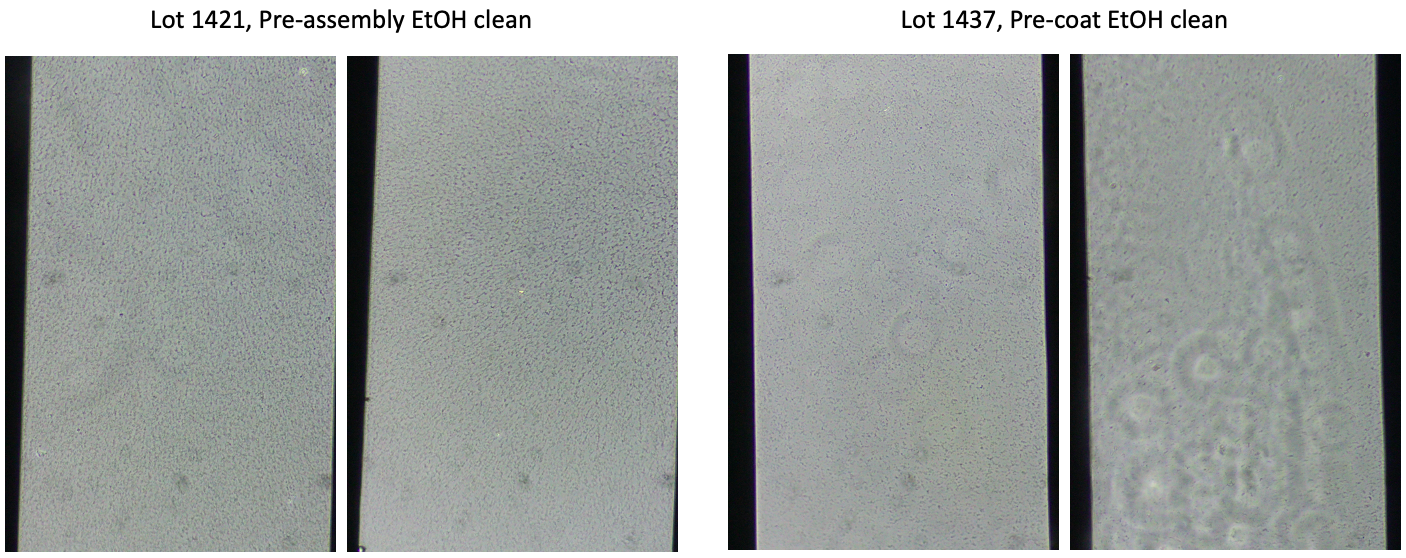
Experiment 8 – Separate Coating Components Test
This is mostly for EECM-BMEC users, but gives more insight into the “black dots”. We put the components (human collagen iv or bovine fibronectin) on separately, as well as the solvent for collagen iv, acetic acid. To make it ‘worse case scenario’, there was no washing in this experiment. The concentrations of each component was stock concentration, so undiluted. I tested both fresh and expired solutions. None of these led to aggregate formation, indicating it is likely an interaction of the two molecules leading to the aggregate formation. Whether this is membrane specific is unclear. This can be alleviated by shorter coating time (4 hr instead of overnight).

Future studies can examine what the reaction is that is happening. We are guessing the fibronectin is coming out of solution, but have not tested this hypothesis.
I also wonder if this is excess coating that forms aggregates because the surface is super-saturated. If true, we could simply used less and they would not show. I know the coating recipes are transferred from experience on cell culture plates, but NPN may have a significantly lower capacity for protein binding compared to plastic.
Thanks for the update Molly. Please add an ethanol clean to all protocols on these pages and in your manuscript.