Diffusion of BSA through SC267 prevented using UV-Ozone
As mentioned in my previous few posts Justin and I have been working to characterize contact angles resultant from various exposures in the UV-Ozone system. Earlier this week we got our new oxygen tank installed and it appears that after using the 15min cleaning protocol (15min@150C with vacuum) noticeable decreases in contact angles can be observed. The path forward is to start performing simple diffusion experiments using rhodamine and fluoroscein to ensure that the contact angle reductions are not a result of the membrane samples being coated with a thin layer of PDMS.
Yesterday I decided to jump one step ahead and ran a SC267 sample in the UV-ozone system for 20min at 150C (after the cleaning protocol). I then ran a diffusion experiment with both this “treated” sample as well as an “untreated” SC267 control. The diffusion experiment was again set up using Jess’ diffusion cell with 6uL of 5mg/mL BSA on the samples topside with 60uL of DI-water on the samples backside.
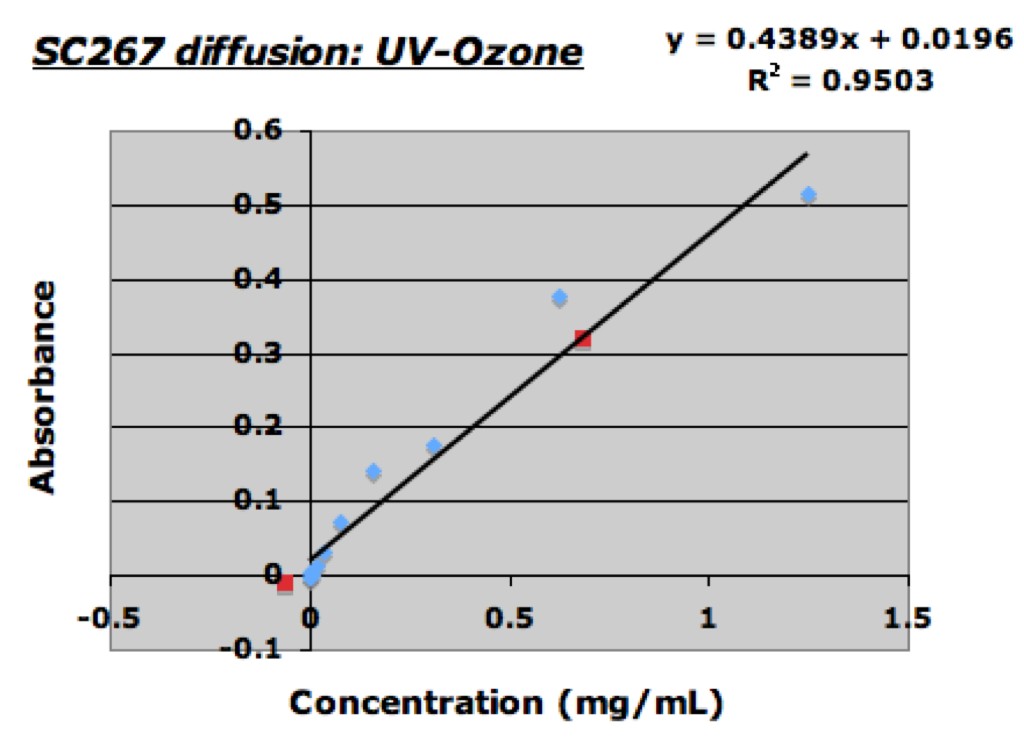
The plot above shows the results of the diffusion experiment. The blue points represent out BSA standards, while the red points represent the “treated” and “untreated” sample filtrates. The “untreated” sample filtrate was calculated to have a BSA concentration of approximately 0.6864mg/mL. On the other hand the “treated” sample filtrate was calculated to have a BSA concentration of -0.061mg/mL (essentially zero). Unfortunately I forgot to take a picture of the assay plate but it was clear to see that the color of the “treated” sample was about the sample as the color of the DI-water controls.
Based on these results we may be preventing the diffusion of BSA by exposing a SC267 sample to 20min of UV-ozone at 150C. The few questions do remain: have we closed the pores as a result of PDMS coverage? If the pores are not closed can DDM diffuse across? In addition to the rhodamine and fluorescein diffusion tests I plan on running a diffusion array comparing “treated” and “untreated” samples with the diffusion of BSA and DDM.
Lets hope this means what we wish it to mean.
Please check the diffusion of positive and negatively charged dyes. If the positive dye passes, bingo.
Have we confirmed that something is being deposited by this system? Haven’t we looked at samples that have been treated in the UV-Ozone system in the TEM? I see no mechanism for this system to deposit anything on the surface, so TEMs should reveal no coverage within the pores. For a deposition process, you need some combination of:
1) a vacuum environment
2) high temperature at some point of vaporization, and a colder substrate for condensation
3) a mix of reactive gases that form a solid product
4) some other fancy way to deliver a film material to the substrate
In this system, I don’t see any of these conditions satisfied, so I’m skeptical. I think this has to be an effect of the ozone oxidizing the surface. It must be affecting the surface charge and/or converting a species that is already in the surface to something else.