Seeing the Softer Side of Silicon: Polymer Coating Silicon Membranes Towards an in vitro Bacterial Infection Mimetic
With our virus sensor paper now published, my journey with virus diagnostics, at least for now, comes to an end. Keeping with the theme of modifying silicon surfaces for interaction with pathogens, I am now switching gears to developing models for bacterial infection. This is currently taking shape in an effort to coat our silicon membranes with soft polymer in order to present a soft surface to Staphylococcus Aureus. This is a part of a larger project which I will outline below. As is laid out, much more work will follow in order to complete the aims of this project.
Project Rationale
The goal of this project is to develop in vitro mimetics of the osteocyte lacuna-canicular network (OLCN) to improve the ability to predict Staphylococcus Aureus invasion and colonization of the OLCN during osteomyelitis (OM). OM is a persistent bone infection occurring during roughly 1% of elective orthopedic surgeries. Despite continuing efforts to improve practices and treatments, reinfection rates have remained at 30% for decades with 10% of cases resulting in sepsis, amputation, or death. The causative agent behind most cases of OM is S. Aureus, an opportunistic pathogen that colonizes the osteocyte lacuno-canilicular network (OLCN) of cortical bone as a means of immune evasion. Because S. Aureus is a ~1 µm coccus with no known motility mechanism, its ability to invade and propagate through the sub-micron spaces of the OLCN is surprising. Previously, a team made up of our lab and several medical center labs made the original discovery of OLCN colonization by S. Aureus in mice and in humans, findings now verified by multiple groups. We also demonstrated the ability of S. Aureus to actively pass through sub-micron pores using an in vitro mimetic we called the µSiM-CA (microfluidic device enabled by a silicon membrane – canalicular array). Recently, we used the µSiM-CA in functional screens and discovered that the cell wall transpeptidase proteins Pbp3 and Pbp4 are both essential for nanopore propagation in vitro. These results were predictive of the inability of Pbp3 and Pbp4 deletion mutants to invade the OLCN in mice. Functional screens in the µSiM-CA are now guiding the discovery of small molecule inhibitors of Pbp3 and Pbp4 as potential prophylactic treatments for OM.
Project Goals
Despite its successful use in identifying essential genes forS. Aureus colonization of the OLCN, the current µSiM-CA is limited in its capacity to provide insight into the full range of mechanisms involved in the phenomena. Notably, the µSiM-CA failed to predict the ability ofS. Aureus mutants lacking the SASC surface adhesion molecule to enter canaliculi in mice. Additionally, two fundamental hypotheses about the biophysical mechanisms underlying OLCN colonization have yet to be directly tested. These hypotheses are: 1) that haptotaxis (migration directed by surface adhesion) and durotaxis (migration directed by environmental rigidity) guide the recognition and invasion of the openings of the OLCN; and 2) that migration is driven by asymmetric cell division so that one daughter cell serves as an anchor while the second propagates into unoccupied regions of the canaliculi. Testing the first hypothesis requires modifications of the µSiM-CA to enable the independent tuning of pore stiffness and adhesion properties. This post regards work towards this effort. We further propose that, once modified to present a more bone-like material toS. Aureus, the µSiM-CA will predict the ability of SASC mutants to enter the OLCN. The second hypothesis requires a redesign of the µSiM-based mimetic so that it contains long sub-micron channels arranged perpendicular to the light path in a microscope. With this configuration, division and propagation ofS. Aureus can be followed in live, super-resolution microscopy. We will approach these goals with the following Aims.
Related to the work in this post
Aim 1. Establish the roles of haptotaxis and durotaxis in S. Aureus invasion of the OLCN. We will modify the porous membranes used in the µSiM-CA so that they assume: 1) stiffnesses ranging from those of soft-tissues to that of silicon nitride (~10X stiffer than bone); and 2) levels of S. Aureus adhesion ranging from ‘non-stick’ to ultra-adhesive (eg. poly-L-lysine). We will then measure the rate of S. Aureus migration from apical to basal compartments of the Transwell™-style devices. We expect our results to reveal a range of permissive and non-permissive values for both adhesion and stiffness such that if either parameter is in a non-permissive regime, transmigration will not occur. We further expect that the adhesive and stiffness values of cortical bone will lie within the permissive ranges.
Related to future work
Aim 2. Develop a µSiM-CA that predicts the ability of SASC mutants to invade the OLCN. We will produce a superior bone mimetic by coating the surface of membranes used in the µSiM-CA with hydroxyapatite and fibronectin. After establishing that the stiffness and adhesiveness of the composite membranes reside in the permissive regimes identified in Aim 1, we will re-test a library of S. Aureus mutants previously used to identify essential genes for OLCN invasion. We expect that the improved mimetic will again predict essential roles for genes that help create motive forces during cell division (PBP3 and PBP4), but will now accurately capture the apparent redundancy the molecules mediating adhesion. Specifically, we expect that the improved bone-mimicking surface will reveal that SASC is not essential for transmigration, correcting a faulty prediction of the unmodified membranes in the original µSiM-CA.
Aim 3. Develop a novel OLCN mimetic to reveal the mechanism of S. Aureus propagation through long sub-micron channels. While the 400 nm thick membranes used in the µSiM-CA provide a model for S. Aureus recognition and entry into the OLCN, they cannot model the subsequent propagation through mm-long canaliculi required for colonization. This leaves basic hypotheses about the mechanisms of OM unaddressed in our in vitro experiments. Specifically, we have hypothesized that the cell division program of S. Aureus is altered by the confinement in canaliculi, possibly including the asymmetric cell devision implied by in vivo images of fixed samples. To address this knowledge gap, we will develop a novel membrane chip for the µSiM platform in which mm-long sub-micron channels are arranged orthogonal to the optical axis of a super-resolution capable microscope. By using intrinsic GFP-markers of division and the cell membrane, we will conduct live-cell microscopy to capture the dynamics of S. Aureus propagation through sub-micron channels; and use small molecule inhibitors and mutants to probe the underling mechanisms.
Coating Silicon Membranes with Polyurethane-Acrylate
Working towards Aim 1 above, we would like to coat the flat side of porous membranes with polymer and etch through the trench side of those coated membranes. In doing so, we would like to develop a chip which on its flat side presents a soft surface that mimics our porous membrane.
First Spin Coating and Etching Attempt
We began by spin coating polyurethane–acrylate (PUA) provided by Dr. Alexander Shestopalov onto 4 window nonporous chips and 3 window 1 µm slit pore chips. The PUA was extremely viscous, so we initially tried spinning at very high speeds. Using a pipette tip, we spread a small amount of polymer across the surface of the chip and spun through the following protocol: step 1 – 1000 rpm for 10 seconds, step 2 – 5000 rpm for 60 seconds. This process gave us fairly uniform coatings on both nonporous and porous chips, but left a large lip on the edge of the chip. We adjusted by increasing speeds and spin times to the following: step 1 – 2500 rpm for 10 seconds, step 2 – 10000 rpm for 300 seconds. After spinning, we baked the chip at 80 degrees Celsius for 10 minutes. Coatings on the nonporous chips cured well, but coatings on the porous chips bubbled up. Analysis of the polymer coating on the nonporous chips showed it was about 6 µm – 15 µm thick, shown below.

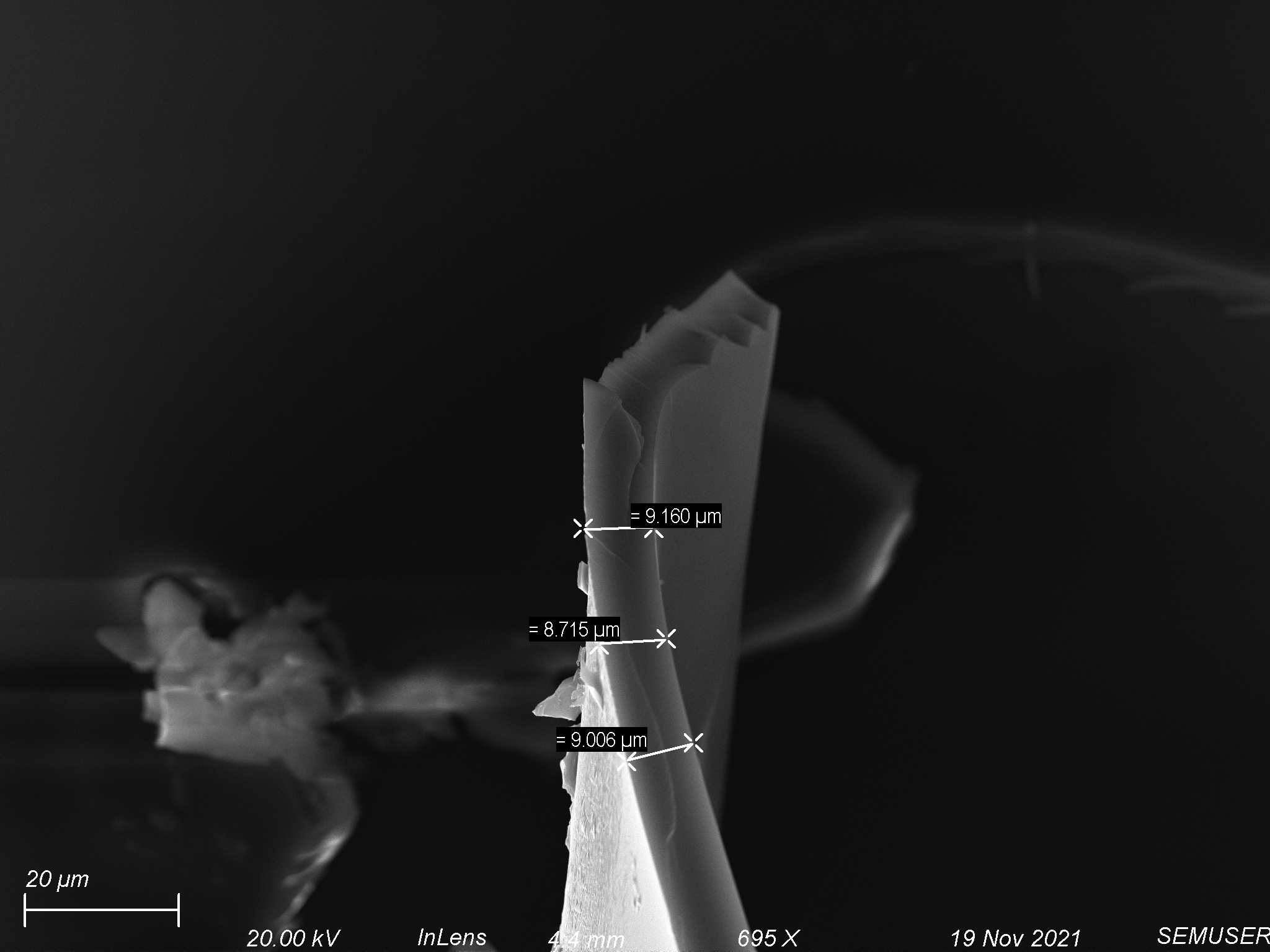

We tried etching the polymer coating on both nonporous and porous chips. A piece of glass was used as a mask on the polymer coated nonporous chips in order to observe the effects of our etching recipe. We used an oxygen/argon etch at 25 scmms each with 150 W of forward power for 10 minutes. We were able to etch most of the polymer off of the nonporous chip, shown below.

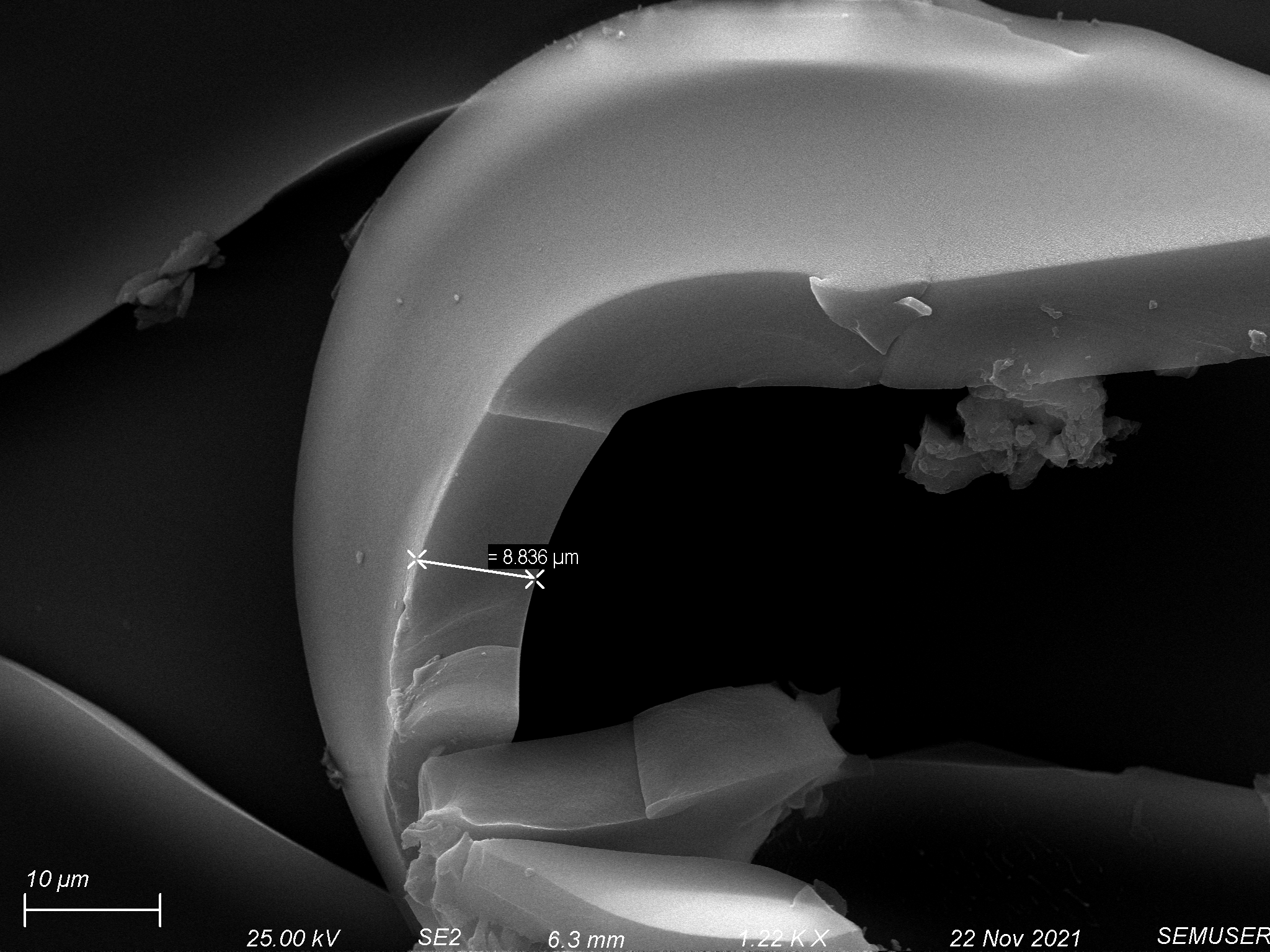
Etching through the polymer coated on the 1 µm slit pore chip, we were able to remove 1-3 µm of polymer while maintaining high fidelity, shown below.



This led us to believe that with the right polymer thickness on a porous membrane, we should be able to etch through the entirety of the polymer layer and maintain porous structure within that layer.
Second Spin Coating and Etching Attempt
Our second approach used the same spin coating protocol as our last attempt in our first approach (step 1 – 2500 rpm for 10 seconds, step 2 – 10000 rpm for 300 seconds). The biggest difference is instead of baking the polymer to cure it, we used UV light to crosslink it instead. We hoped that by using this approach we would get the coating on the porous chips to remain flat. Fortunately, when we UV cured the deposited polymer for 30 minutes, this was the case. Analysis of these polymer coatings showed 8 um thick polymer coatings, shown below.

Etching with the same recipe used in approach 1 (oxygen/argon etch at 25 scmms each with 150 W of forward power for 10 minutes) seemed to take off 2-3 µm of polymer, shown below.



We also tested how well our etch recipe would remove polymer through the pores of a membrane using a 0.5 µm slit pore chip as a shadow mask (by placing a 0.5 um slit pore chip flat side down on top of the polymer coating deposited on nonporous chips). The results were encouraging, showing deep pores that maintain high fidelity. The results of this approach are shown below.





Third Spin Coating and Etching Attempt
For our third approach, we tried warming the PUA slightly to make it less viscous. At room temperature, it did not seem much less viscous. Despite this, we tried the same spin coating recipe as before and got very similar results shown below.
 In an attempt to get more anisotropic etching into our polymer, we modified our etch recipe slightly. For this round we used an oxygen/argon etch at 12.5 sccms each with 150 W forward power. We used 0.5 µm slit pore shadow masks, but also tried 0.5 µm hole pore shadow masks. We again saw the removal of around 3-4 µm of polymer, but with greater fidelity and pattern transfer. This is shown below.
In an attempt to get more anisotropic etching into our polymer, we modified our etch recipe slightly. For this round we used an oxygen/argon etch at 12.5 sccms each with 150 W forward power. We used 0.5 µm slit pore shadow masks, but also tried 0.5 µm hole pore shadow masks. We again saw the removal of around 3-4 µm of polymer, but with greater fidelity and pattern transfer. This is shown below.









Coating Silicon Membranes with Polydimethylsiloxane (PDMS)
To see if a polymer with less viscosity would coat the membrane closer to our desired thickness (1 µm), we tried coating nonporous chips with 10:1 monomer:hardener PDMS. This was degassed and deposited onto the membrane using the same spin coat procedure as before (step 1 – 2500 rpm for 10 seconds, step 2 – 10000 rpm for 300 seconds). This was cured via baking on a hotplate at 210 degrees Celsius for 10 minutes creating a roughly 1 µm thick coating of PDMS, shown below.

To etch the PDMS, we used an oxygen/sulfur hexafluoride etch with both at 15 sccms using 125 W forward power for 10 minutes. This created rougher looking hole pores, but transferred the pattern fairly well, shown below.



Conclusions
Overall, we have made progress towards the deposition of a 1 µm thick soft polymer on our silicon membranes. While the PUA layers are still too thick, we plan to attempt to thin the polymer to allow it to spread better. PDMS shows us that a thinner polymer can be deposited at the thickness that we desire. Separately, the etching that we have done has shown that we should be able to etch through a soft polymer with a thickness of 1 µm while maintaining high fidelity using the recipes that we have developed up until this point.