Discoloration – Cell Media
I’ve been learning the discoloration protocol from Karl and Anant and finally have some pictures of my own to show. We got a couple of new media (RPMI and DMEM/F12) last week in order to grow the cells I’m getting from the Elder lab (rat lung cells). I did a discoloration test to see if these new media were less harsh than the DMEM and EGM currently used by Karl and Anant. All media had 10% FBS, all samples were in the incubator and these are from untreated wafer 336. I apologize for not having the same format as Karl:
This clearly shows that DMEM is the harshest media, causing discoloration in less than a day. Interestingly, the membranes show no discoloration after 2 days in L15 only. I did a quick comparison between the media components. The major differences between DMEM and L15 are that: DMEM has much more sodium bicarbonate than all other media, and it is the only media that lacks sodium phosphate dibasic. The 2 unique components of L15 are sodium pyruvate and potassium phosphate monobasic. The differences in Na bicarbonate, Na phosphate and K phosphate point to buffering capacity (pH-mediated discoloration?), but Na pyruvate can protect against H2O2-mediated oxidative stress (oxidation-mediated discoloration?).
I also monitored the pH of these media types over 2 days in the incubator. I only followed the pH for 2 days because this is the typical interval that media is changed for cell culture (at least in my experience). This is n=2 experiments with standard deviation as error bars.
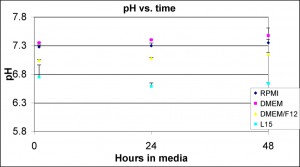
This data supports Chris’s idea that discoloration is pH-driven since DMEM has a pH about 0.6 units higher than L15. Unfortunately, it’s not proof.

Were all these studies done in the incubator?
My current opinion is that this effect is purely pH based. Would the pH of the L15 buffer in the incubator be lower than the other media, given the low bicarb concentration? HIgh pH dissolves Si, low pH does not.
I suppose there may be other oxidative chemistries that may also contribute, but nothing has been confirmed yet.
If we could get pH measurements in all these solutions over time, this may become more clear.
Are these really big enough differences in pH to explain the entire story?
If it is strictly pH we should be able to do this in a standard buffer with a slightly basic pKa – like TRIS and see the membranes dissolve in the incubator faster than say MES which has a slightly basic pKa. Be sure to monitor the pH and make sure the starting mixtures are at the pKa of the buffers.
We did this before and saw nothing, but we didn’t put the membranes in the incubator.