SC256 Diffusion with a Mixture of BSA and DDM
In an effort to conserve membrane samples I am working to run diffusion experiments with a mixture of BSA and DDM. This will only use 2 chips instead of 4. In our meeting on Tuesday it was mentioned that BSA can be detected using the TECAN at 280nm, while DDM is not detected at that wavelength. I ran a quick test to make sure and the image below shows that at 280nm BSA concentration can be measured in a mixture of BSA and DDM. In addition, back at the beginning of the summer I showed that the DDM assay does not pick up BSA.
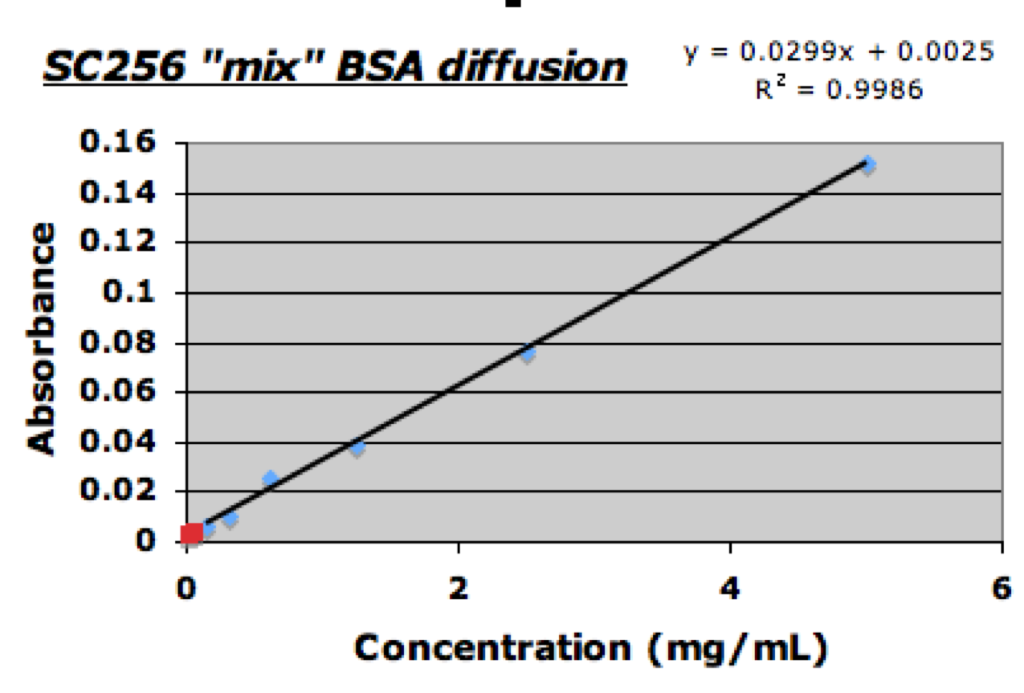
Yesterday I set up a diffusion experiment to tests “treated” and “untreated” SC256 samples with the diffusion of a mixture of BSA and DDM in 20mM HEPES at 7.5pH. This morning I collected the filtrates and used 3uL of each with the NanoQuant TECAN plate to determine the BSA concentrations beore running the DDM assay.
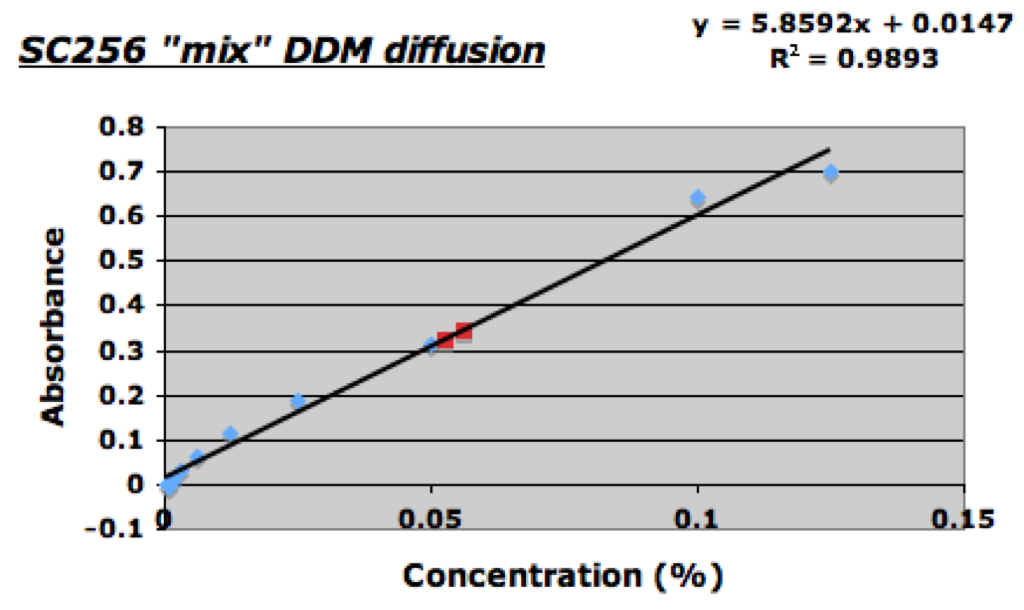
From the analysis using a standard BSA absorbance curve it was determined that the “untreated” filtrate concentration was ~0.06mg/ml; the “treated” filtrate concentration on the other hand was calculated to be ~0.018mg/mL. I am not sure if this decrease in concentration is a result of the UV-Ozone treatment as both are well below the initial retentate concentration of 2.5mg/mL. I plan on setting up another diffusion experiment today so on monday I might have a better idea. Once I had measured the BSA absorbance I was able to perform the usual DDM assay with the rest of the collected filtrate samples. As shown below both the “treated” and “untreated” SC256 samples allowed for diffusion of DDM.
The calculated DDM concentration of the “untreated” SC256 sample was ~0.056%, and the “treated” sample was ~0.052%; relatively equal.


What was the starting concentration of DDM? What volumes are you working with in the filtrate and retentate?
The starting concentration of DDM was 1.0% and of BSA was 2.5mg/mL. The setup had an initial retentate volume of 6uL and an initial filtrate volume of 60uL.