Endothelial Focal Adhesion BMES Abstract 2015
The following post is the abstract that I recently submitted for the BMES Annual Conference.
Introduction: Permeable membranes provide a physiologically relevant cell culture substrate for in vitro study of barrier cell types, such as endothelial cells, which assume a polarized morphology with distinct apical and basolateral surfaces. Cell morphology and subsequent monolayer formation and barrier function are largely dependent on initial interactions with the culture substrate, specifically adhesion. Cells form contacts with the cell culture substrate via focal adhesions. Previous studies have shown that cells fully embedded within a 3D matrix will not form focal adhesions, while cells cultured on 2D substrates will form focal adhesions. We hypothesize that a highly permeable, ultrathin membrane could act like a “slice” of a 3D matrix. We tested this hypothesis by investigating the formation of focal adhesions on optically transparent SiO2 membranes with a variety of pore sizes (0.5-5.0 μm) and porosities (LP, 5.7% and HP, 19.6%).
Materials and Methods: SiO2 membranes were fabricated using standard MEMS processing techniques. 300 nm of SiO2 was deposited using PECVD and annealed to create a tensile film on polished silicon wafers. Both device geometry and pore structure were patterned using common photolithography techniques. Pores were formed using a reactive ion etch. Membranes were fabricated with 0.5 μm, 3.0 μm, and 5.0 μm pore sizes with 5.7% or 19.6% porosity. Tensile and standard compressive SiO2 films were compared in strength using burst pressure tests. Human umbilical vein endothelial cells (HUVECs) were cultured on the porous SiO2 membranes, as well as non-porous SiO2 and tissue culture plastic (TCP). Cells were stained using phalloidin and anti-vinculin antibody in order to visualize F-actin and vinculin, respectively, via fluorescence microscopy.
Results and Discussion: The presence of vinculin in focal adhesions in HUVECs cultured on porous SiO2 membranes was comparable to that of non-porous SiO2 membranes and TCP. This shows that HUVEC respond to the SiO2 membranes as 2D substrates.

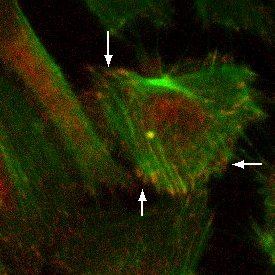
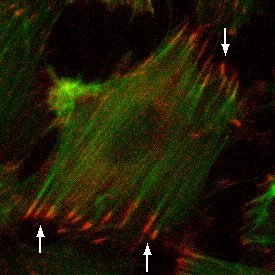
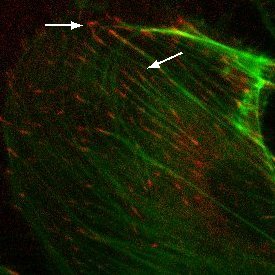
Conclusions: HUVEC form focal adhesions on porous SiO2 membranes equivalent to focal adhesions observed in HUVEC cultured on non-porous SiO2 membranes and TCP. Future studies will investigate nanoporous membranes of similar porosity to determine if minimal contiguous contact area inhibits focal adhesion formation in HUVEC. Additionally, we are investigating whether permeable substrates influence barrier formation in an endothelial cell monolayer compared to non-porous 2D controls.