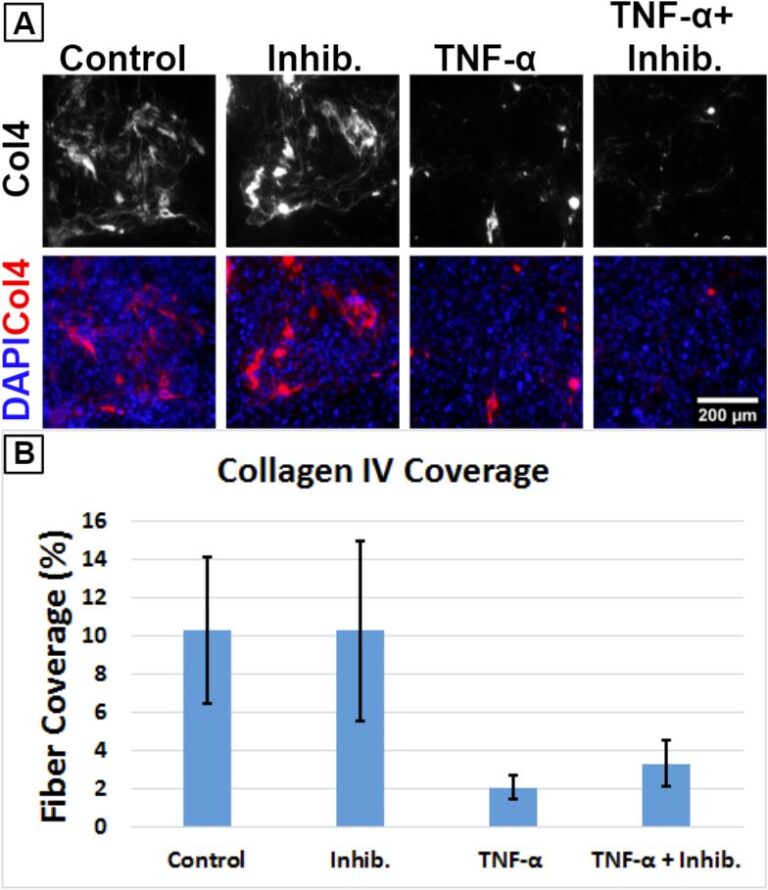
Assessing Neutrophil Activation inside a Flow Cell Device with Flow Cytometry
Introduction As part of our continuing evaluation of the flow cell developed by our collaborators at RIT, our next goal is to demonstrate that neutrophils remain inactivated when flown through a first generation device (Figure 1). For this, we studied neutrophil activation as a function of fluid flow induced shear stress based on the physical…